A new technique called micro-optical coherence tomography enhances percutaneous coronary interventions to allow the study of atherosclerosis at the cellular level.
RADHIKA K. PODUVAL AND GUILLERMO J. TEARNEY, WELLMAN CENTER FOR PHOTOMEDICINE
Optical coherence tomography (OCT) is an interferometric imaging technique that uses light in the near-infrared spectrum to provide detailed, cross-sectional images of biological tissue. OCT is therefore widely used for clinical ophthalmology and has potential for minimally invasive microscopic imaging of other organ systems in vivo. For interventional cardiology, implementing OCT using an intracoronary catheter enables the imaging of coronary morphology in real time in vivo1. The spatial resolution of OCT at ~15 μm is an order of magnitude higher than predicate intravascular ultrasound, enabling the visualization of more detail pertaining to arterial atherosclerotic pathology.
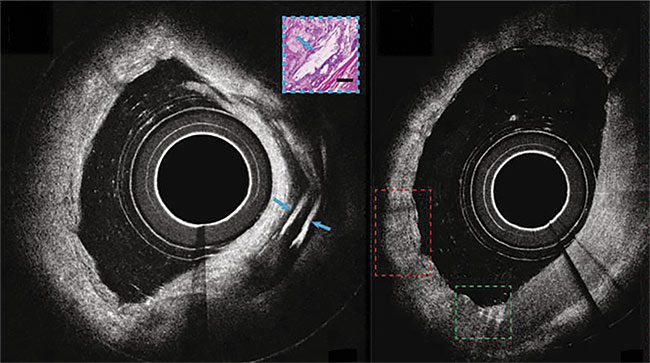
Micro-OCT views of coronary arteries with cholesterol deposits becoming visible. Adapted with permission from Reference 8.
OCT is currently used in clinical settings to guide the selection of stent parameters, such as length and diameter, and to confirm the appropriate stent placement during percutaneous coronary interventions. Multiple studies have shown that the utilization of OCT to optimize stent placement improves patient outcomes, allowing for proper arterial functioning and blood flow2. In addition, OCT has been proposed to identify atherosclerotic lesions at risk for progression and future thrombosis, or clotting2. This latter capability may make it useful in the guidance of stenting high-risk limiting lesions during percutaneous coronary interventions.
Intracoronary imaging is not yet used to guide the treatment of lesions that do not limit blood flow. This lack of use stems from the relatively low precision of the prevalent intracoronary imaging modalities in predicting future coronary events, due to limitations in imaging resolution. Among these, OCT has the highest imaging resolution. However, a missing component in conventional OCT systems, that decreases specificity for high-risk plaques, is the inherent inability to visualize individual cells such as endothelial cells, macrophages, and smooth muscle cells, along with subcellular structures (platelets, fibrin, crystals, and microcalcifications). These cell types are associated with the pathogenesis of atherosclerosis progression and lesion destabilization.
Micro-OCT technology
Interventional cardiologists are highly interested in studying the mechanism of atherosclerosis and the markers of its onset using a high-resolution imaging technology such as OCT. However, the resolution of conventional systems is inadequate for imaging coronary atherosclerotic pathobiology characterized by cells, such as fibroblasts, smooth muscle cells, monocytes, macrophages, and neutrophils. But, recently, a new form of OCT has been developed that enables the higher-spatial-resolution visualization of human tissue between 1 and 2 μm. This technique, termed micro-OCT, enables the study of atherosclerosis at the cellular level. The image quality and level of detail visualized using micro-OCT compared to conventional OCT is greatly improved (Figure 1).
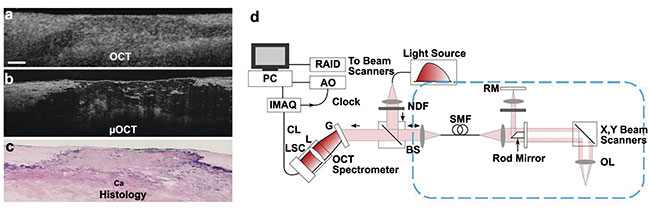
Figure 1. Micro-OCT images of fibrocalcific human cadaver coronary plaque, comparing corresponding OCT (a), micro-OCT (b), and histology (H&E of a calcium plate within the coronary artery wall) (c). A schematic of a benchtop micro-OCT system (d). Scale bar: 200 µm. Adapted with permission from Reference 3.
Micro-OCT was first demonstrated within a benchtop microscopy system, in which the technique was shown to be capable of visualizing many key cellular and subcellular features relevant to atherogenesis, plaque rupture, thrombosis, and neointimal healing after stenting3. Although single scans are tomographic images, micro-OCT data can be obtained in 3D by scanning the sample arm optics along the tissue of interest to create a scaled volumetric reconstruction (Figures 2 and 3). The advantages of 3D micro-OCT for characterizing atherosclerosis are profound because many more biomarkers that are relevant to plaque progression can be identified and characterized in three dimensions, compared to two-dimensional histology.
A micro-OCT system is composed of a spectral domain OCT system (SD-OCT) that includes a Michelson interferometer in a common-path reference configuration (Figure 1d). The source is typically a supercontinuum laser in the 600- to 900-nm NIR wavelength range, for instance 800 ± 150 nm, compared to traditional cardiac OCT systems that have longer central wavelengths in the range of 1250 to 1350 nm. The spectrometer and sample arm optics in micro-OCT are optimized for 1- to 2-μm axial and lateral resolution through extended spectral bandwidths and high NA sample arm optics with depth of focus extension respectively. This improved resolution compared to conventional OCT results in cross-sectional tissue imaging with significantly higher quality and detail (Figures 1a-c).
This technology has been further developed and miniaturized during the last decade, replacing the sample arm with optical fiber-based imaging catheters containing high numerical aperture micro-optical components. Micro-OCT technology has recently proved to be effective in subsurface cellular resolution pulmonary, gastrointestinal, cochlear, and coronary imaging. Combined with rotational pullback imaging — or helical scanning, which combines the translation of the imaging device with simultaneous rotation along the central axis — micro-OCT can provide the visualization of cells and subcellular features along the radial depth of luminal organs such as coronary vasculature.
Cellular-level imaging
Micro-OCT has proved to be capable of imaging key markers of early atherogenesis, plaque progression, and high-risk atheromas. This includes changes in endothelial cell patterning on the coronary artery surface, such as the presence of monocytes and macrophages, neutrophils, phagocytosing immune cells, crystals (cholesterol or urea), and calcium plates within the plaque.
The imaging of coronaries using micro-OCT has revealed unique insights into the importance of surface morphology and the thickness of endothelial cell coverage in the innermost layer of the coronary vessel wall, or intima, to plaque vulnerability4. Endothelial cells are organized in a typical cobblestone pattern, called endothelial pavementing, on the intimal surface5. Disruption of this pattern has been observed by researchers to initiate coronary atherogenesis because it allows low-density lipoprotein and leukocytes to pass into the intima, leading to plaque formation5. Changes in the morphology of the intimal surface can provide key insights into the histopathological precursors of acute coronary syndrome.
Scanning electron microscopy (SEM) is currently the gold standard for endothelial tissue imaging ex vivo. But SEM requires complex sample preparation with imaging in a vacuum chamber, rendering it unsuitable for either fresh tissue imaging or in vivo imaging. Micro-OCT, however, requires negligible tissue preparation for ex vivo imaging, it allows preservation of tissue orientation, and it can facilitate the study of vascular flow-induced changes in the endothelial coverage pattern and thickness6. Further, the micro-OCT images that were obtained from the coronary arteries of an atherosclerotic human cadaver artery showed that endothelial cell coverage was prominent over healthy coronary segments with intimal thickening, whereas coverage was lacking in lesions with superficial nodular calcification with a necrotic core (Figures 2a and 2c)7.
Further, the endothelial cell coverage over the fibroatheromatous plaque was intermediate (Figure 2b), but significantly lower than in healthy coronary arteries. The surface coverage was quantified through intimal surface roughness characterization from the micro-OCT images, revealing significant differences in fibroatheromatous (composed of cholesterol deposits) and fibrocalcific (composed of calcium) plaques compared to less severe atherosclerotic lesions4. Moreover, it was observed that apoptotic macrophages, a class of immune cells, were not efficiently cleared in high-risk, thin-cap fibroatheroma lesions, thus becoming part of the plaque buildup. This feature can make the thin-cap fibroatheromas vulnerable to secondary necrosis with fibrous cap thinning and enlargement of the necrotic core. Intravascular intimal surface roughness measurement through micro-OCT during percutaneous coronary interventions could potentially be a novel marker of early coronary atherosclerosis and could pave the way to establishing quantification metrics for plaque vulnerability.
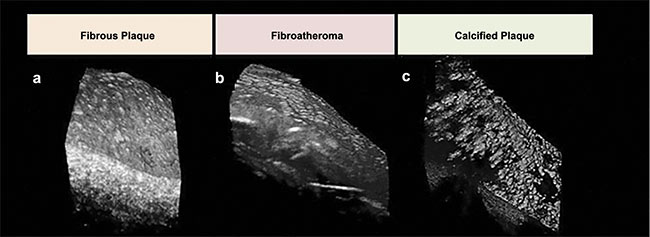
Figure 2. Representative micro-OCT images for varying plaque morphologies. Compared to the lesion with intimal hyperplasia (a), endothelial cells over
fibroatheroma (b) and fibrocalcific plaques (c) are sparse. Adapted with permission from Reference 6.
The visualization of vascular inflammation can help improve our understanding of the mechanism of atherosclerosis progression, to guide therapy in treating and managing coronary artery disease. Neutrophil adhesion to the coronary endothelial surface and subsequent monocyte and macrophage infiltration plays a key role in the progression of atherosclerotic lesions (Figure 3). Micro-OCT was proved capable of identifying specific inflammatory cells ex vivo in human coronaries. Monocytes are visualized as large cells with scant cytoplasm and bean-shaped nuclei adhering to the intimal surface (Figure 3d). Macrophages are present in images as large, flocculent, round, or ellipsoidal cells that are highly scattering, as confirmed by micro-OCT images of cultured macrophages in vitro. Cholesterol phagocytosis (ingestion) of cholesterol crystals by macrophages causes elevated cytoplasmic scattering in micro-OCT images of macrophages, as seen in the 3D-micro-OCT image of individual macrophages phagocytosing highly scattering cholesterol crystals. Leukocytes adhering to the coronary endothelial surface have also been imaged by micro-OCT. Pseudopods of these inflammatory cells highlight anchor sites to the endothelium, as seen in both the 3D micro-OCT image and the corresponding SEM image. The ability of micro-OCT for noncontact visualization of cellular features suggests a potential for next-generation coronary imaging to enhance our mechanistic understanding of coronary artery disease.
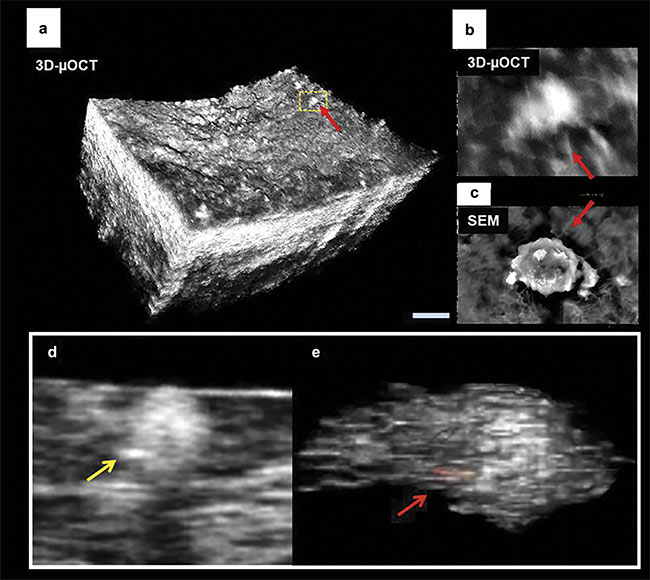
Figure 3. 3D micro-OCT of endothelial cell morphology of a lipid-rich human coronary plaque with adhered leukocytes (red arrows) (a, b). Co-registered scanning electron microscopy (SEM) image showing an individual leukocyte tethered to the surface (c). A representative image of macrophage cells in a human coronary artery containing highly scattering inclusions within their cytoplasm (e). Scale bars: 100 µm (a); 10 µm (b, c). Images (a) and (c) adapted with permission from Reference 6; images (d) and (e) adapted with permission from Reference 7.
Intravascular catheters
Micro-OCT has evolved from its initial integration into a benchtop microscopy system to devices dedicated to enable percutaneous luminal imaging in vivo. Intravascular micro-OCT catheters have been demonstrated for conducting ex vivo imaging of human cadaver coronary arteries and in vivo imaging of atherosclerotic rabbit aorta8. Circumferential micro-OCT views, akin to conventional intravascular OCT and intravascular ultrasound systems, can display vascular smooth muscle cells in lesions with intimal thickening and cholesterol crystal accumulation in human fibro-atheromatous plaques ex vivo (Figure 4). Cross-sectional micro-OCT allows for clear visualization of macrophage movement4.
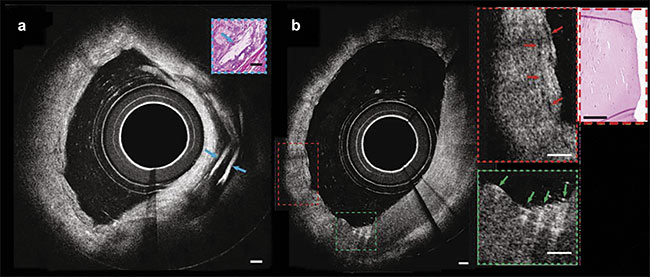
Figure 4. Images from a catheter-based intravascular micro-OCT of a human cadaver coronary ex vivo (a). A circumferential view of micro-OCT showing
multiple cholesterol crystals characterized by strong optical reflections from their top and bottom surfaces, and a cross section of an artery exhibiting probable smooth muscle cells (red arrows) (b, inset) and macrophages undergoing diapedesis (green arrows) (inset). Scale bars: 100 µm. Adapted with
permission from Reference 8.
The key modifications to a micro-OCT system that are required to enable intravascular imaging include an optical fiber-based sample arm catheter with high numerical aperture micro-optics and a compact interferometer implemented as a cart-based system suitable for preclinical and clinical surgical environments. The specialized optics requirements for micro-OCT to achieve higher resolution with increased depth of field with miniaturized instrumentation pose challenges to the scalability and repeatability of imaging performance. These issues can potentially be addressed by using ongoing innovations in 3D nanofabrication for high-precision optomechanical fabrication using two-photon polymerization lithography. Some preliminary developments in this field have been demonstrated for conventional OCT resolution and more recently for micro-OCT of coronary arteries.
Micro-OCT shows great promise for improving clinical care in observing plaque initiation, plaque development, and for potentially guiding personalized intervention based on the diagnostic capability of cellular and subcellular plaque morphology. Catheter-based micro-OCT has been shown to be capable of imaging individual crystals and inflammatory cells not only ex vivo in human coronary plaques but also in animal models in vivo. Grant proposals are currently being reviewed for potential expansion into clinical trials. Percutaneous coronary interventions-based observations of tissue morphology and the cellular-level details visualized by intravascular micro-OCT catheters in humans could potentially help improve clinical risk classification metrics for atherosclerotic plaques — for instance, based on the endothelial surface-roughness quantification.
Micro-OCT also holds promise in targeting individualized therapy for existing blockages based on observed cellular biomarkers. Micro-OCT imaging in percutaneous coronary intervention catheters could potentially enable targeted individualized drug delivery based on local pathology within the vasculature, and by monitoring its efficacy by tracking localized cellular changes in the artery wall in response to the drug treatment. Observation of coronary cells in vivo during percutaneous coronary interventions, in concert with the longitudinal monitoring of the dynamic process and progression of atherosclerosis, therefore has the potential to promote research and clinical protocols in cardiology for years to come.
Meet the authors
Radhika K. Poduval, postdoctoral research fellow at the Wellman Center for Photomedicine within Massachusetts General Hospital and Harvard Medical School, Boston, is working on miniaturizing fiber-optic endoscopic devices. She received a doctorate degree in electronic and electrical engineering from University College London; email: [email protected].
Guillermo J. Tearney, M.D., Ph.D., is the Remondi Family Endowed MGH Research Institute Chair, professor of pathology at Harvard Medical School, and an affiliated faculty member of the Harvard-MIT Division of Health Sciences and Technology. He maintains his lab at the Wellman Center for Photomedicine at the Massachusetts General Hospital. Tearney received his Doctor of Medicine degree magna cum laude from Harvard Medical School and his doctorate in electrical engineering and computer science from the Massachusetts Institute of Technology; email: [email protected].
Acknowledgments
This work was supported by the U.S. National Institute of Health grant R01 HL076398 and Verdure Biotech.
References
1. B.E. Bouma et al. (2003). Evaluation of intracoronary stenting by intravascular optical coherence tomography. Heart, Vol. 89,
No. 3, pp. 317-320.
2. H. Sinclair et al. (2015). OCT for the identification of vulnerable plaque in acute coronary syndrome. JACC: Cardiovasc Imaging, Vol. 8, No. 2, pp. 198-209.
3. L. Liu et al. (2011). Imaging the subcellular structure for human coronary atherosclerosis using 1-μm resolution optical coherence tomography (μOCT). Nat Med, Vol. 17,
No. 8, pp. 1010-1014.
4. K. Nishimiya et al. (2019). Micro-optical coherence tomography for endothelial cell visualization in the coronary arteries.
JACC Cardiovasc Imaging, Vol. 12, No. 9,
pp. 1878-1880.
5. P.M. Vanhoutte et al. (2017). Endothelial dysfunction and vascular disease — a 30th anniversary update. Acta Physiol, Vol. 219, pp. 22-96.
6. K. Nishimiya et al. (2023). OCT emerging technologies: coronary micro-optical
coherence tomography. Interv Cardiol Clin,
Vol. 12, No. 2, pp. 237-244.
7. M. Kashiwagi et al. (2014). Feasibility of the assessment of cholesterol crystals in human macrophages using micro optical coherence tomography. PLoS One, Vol. 9, No. 7,
p. e102669.
8. B. Yin et al. (2019). 3D cellular-resolution imaging in arteries using few-mode interferometry. Light Sci Appl, Vol. 8, p. 104.