Technology expands upon quantitative phase imaging, which can be integrated into microscopes commonly found in the laboratory.
BJÖRN KEMPER, BIOMEDICAL TECHNOLOGY CENTER, UNIVERSITY OF MÜNSTER
In recent years, quantitative phase imaging (QPI) has been continuously improved for purposes of high-resolution, label-free quantitative microscopy1. Label-free imaging has received increased attention in the last decade as a minimally invasive way to observe proteins and cells, and to study the behavior and properties of biological specimens with minimized modification and sample preparation. Using noninvasive techniques means samples under investigation are not influenced by fluorophores or dyes, which can change the physiological processes or behavior of samples by causing cellular motility or migration. Moreover, because QPI requires only low light intensities for object illumination, the potential harm light could cause to a sample is minimized. Low light is a precondition for the long-term monitoring of living cells because high doses of light radiation can cause cell death or photo damage.
Digital holographic microscopy (DHM)2,3 is an interferometry-based variant of QPI that typically uses a laser as a coherent light source and provides QPI by detecting specimen-induced optical path length changes against the surrounding environment. DHM can be modularly integrated into common optical microscopes3,4, which allows its integration as a label-free imaging modality in research laboratories.
In DHM, reconstruction of digitally captured holograms is performed numerically by a computer. Thus, multifocus
imaging of specimen parts in various layers is achieved — and even subsequent
autofocusing from single captured digital holograms — without the need for mechanical focus realignment5. This is advantageous for the correction of unpredictable focus drifts during long-term observations of living cells, or for imaging of cell movements in 3D environments such as collagen or gel matrices. In addition, DHM-based phase contrast imaging can simplify automated object tracking and image segmentation for quantification of cell migration and morphology by extraction of absolute biophysical parameters such as cellular volume, thickness, and dry mass1. Moreover, the evaluation
of quantitative DHM phase images provides access to optical parameters such as the cellular refractive index, which is related to the cellular water content, intracellular solute concentrations, and tissue density3,6. This qualifies DHM as a viable label-free cytometry tool for the identification and characterization of tumors, blood, and stem cells in mixtures or composites; for time-lapse in vitro toxicity and drug testing; and for ex vivo histology of dissected tissues.
QPI with DHM
Figure 1 illustrates the concept of a typical laser-based off-axis setup for QPI with DHM2 that can also be integrated into commercial research microscopes3,4 for use in a biomedical laboratory. An inverted transmission arrangement allows bright-field imaging and DHM investigations on mainly transparent samples, such as unstained tissue sections or living cell cultures in a petri dish filled with cell culture medium. Light from single-mode, TEM00, solid-state lasers with long coherence length (e.g., a Cobolt 06-DPL or 08-DPL 532-nm laser operating at 25 or 50 mW output power and achieving more than 100 m of coherence length) is divided into an object illumination wave O and a reference wave R. While O passes the sample, the undisturbed wave R is guided directly but slightly tilted to the digital image recording device, such as a CCD or CMOS sensor. From the recorded interference patterns (digital off-axis holograms), quantitative phase images can be numerically reconstructed2,5 that quantify the optical path length changes induced by the morphology and refractive index distribution of the investigated specimen against the surrounding medium2, and from which biophysical cell and tissue parameters can be extracted3-7.
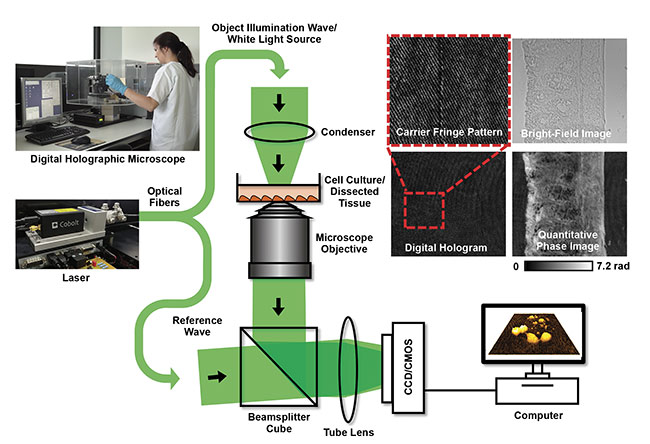
Figure 1. The concept of laser-based inverted off-axis DHM in transmission configuration for correlated label-free bright-field and quantitative phase imaging of dissected tissues (upper right) and living cell cultures in the environment of a biomedical laboratory. Adapted with permission from References 3 and 4.
Identifying cells and culture quality
DHM can be used to distinguish among various cell types; for example, to identify specific tumor cells in co-cultures or blood samples without labeling1,7. Figure 2a shows representative QPI images and refractive index data retrieved from two different confluently grown pancreatic tumor cell lines originating from the same donor, with the data plotted against the cell radius. For the measurements, cells were detached and observed with DHM in a suspension with physiological osmolality. Because of the spherical appearance of most detached cells, the refractive index and radius can be acquired by 2D numerical fitting procedures from quantitative phase images of individual cells. In the same process, the cellular dry mass can also be determined. The dot plot in Figure 2a shows an example of a fraction of n = 100 investigated cells for each cell type. The two cancer cell types can be distinguished clearly by the cell radii as well as their refractive indices.
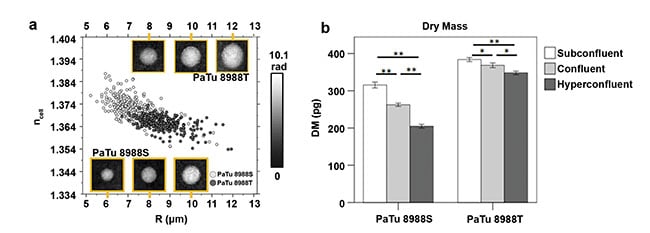
Figure 2. Refractive index ncell of detached pancreatic tumor single cells (PaTu 8988T, PaTu 8988S) plotted versus the cell radius (R). Inserts show representative quantitative DHM phase images of the various cell types (a). The impact of various growth conditions (subconfluent, confluent, hyperconfluent) on the average cellular dry mass (DM) of PaTu 8988S and PaTu 8988T cells (b). Samples of each condition were analyzed in N = 3 independent experiments in which a number of n = 100 cells were analyzed, respectively. *,**Statistical significance; pg: pictograms. Adapted with permission from Reference 7.
DHM also enables the quantification of cell alterations caused by culturing conditions by determining biophysical cell parameters. To demonstrate this capability, tumor cells that had been cultivated in various densities7 — including subconfluent, confluent, and hyperconfluent — were analyzed. As shown in Figure 2b, a significant change of the single cell average dry mass was detected for the two observed cancer cell types. This shows that DHM reproducibly can quantify the cellular response to various growth and environmental conditions and thus verify the consistency of cell cultures, an essential parameter in all cell-based biomedical assays.
Label-free in vitro toxicity testing
QPI with DHM has the potential to be used as a
powerful routine tool in various areas of biomedicine and toxicity testing, such as in cell biology, cancer research, nanotoxicology, and histopathology.
Common in vitro toxicity tests of drugs, chemicals, or nanomaterials involve the measurement of cellular end points — such as stress response, cell viability, proliferation, or cell death — to determine, for example, the effectiveness of anti-cancer drugs or the harmfulness of environmental pollutants or pathogens. Many current assay systems are based on enzyme activity or protein expression by optical readout of enzyme substrates or marker protein labeling. Such procedures have to be stopped at distinct time points for readout and can require several time-consuming sample preparation steps. In such scenarios, DHM offers simplification by continuous label-free monitoring of growth and morphology changes. Figure 3 shows results from human colon carcinoma cells exposed to toxic vesicles and observed during time-lapse investigations versus untreated control cells8. Digital holograms of the cells were acquired (with a setup as shown in Figure 1) continuously every 3 min for 48 h. To quantify cellular growth and morphological changes, the resulting series of quantitative phase images were evaluated by image segmentation for the area occupied by the cells and the cell-induced average phase contrast from which subsequently the cellular dry mass was also calculated. Control cells displayed regular growth, resulting in an increase of the cell-covered area, cell density, and cell layer thickness (left column of Figure 3a and upper panel of Figure 3b), while cells treated with toxic vesicles moved out of the cell aggregate and underwent apoptosis (right column of Figure 3a and lower panel of Figure 3b). This is reflected by the simultaneously retrieved temporal development of the cellular dry mass (Figure 3c). In summary, the results in Figure 3 demonstrate the capability of DHM for effective in vitro toxicity testing without additional staining or absorption/fluorescence-based readout. QPI is different than specific marker-based screening in that it measures whole cell alterations. This allows the study of the entire impact on specimens during the development of new drugs, such as for cancer treatment. In toxicology, interferometry-based techniques such as DHM could also improve the observation of the very unpredictable toxic effects of nanomaterials that are released into the environment, because these particles often exhibit optical properties, such as absorbance or fluorescence, that disturb the established assays3.
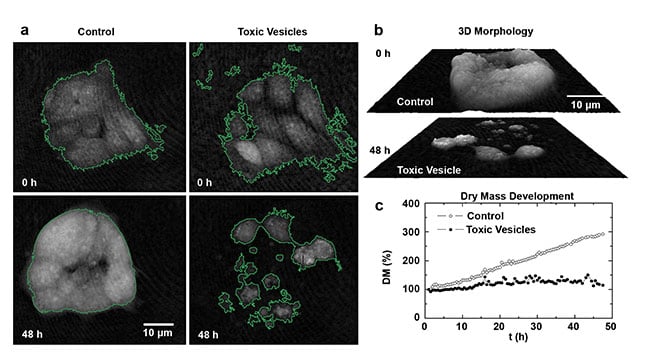
Figure 3. DHM monitoring of the cellular response to toxins. Segmented (green outlines) quantitative DHM phase images of human colon
carcinoma cells (HCT-8) at t = 0 h and 48 h after incubation with only cell culture medium (control) or with toxic vesicles (OMV LB226692) (a).
Illustration of the cell morphology by pseudo 3D representations of the quantitative DHM phase contrast images in (a) after 48 h (b). Dynamics
of cellular growth, quantified by the normalized temporal course of the cellular dry mass (DM) (c). Adapted with permission from Reference 8.
Quantification of inflammation
QPI with DHM can also be used for assessment of dissected tissues ex vivo. For example, it can be used to measure inflammation-mediated morphological alterations6,9 in chronic inflammatory bowel diseases (IBD), such as Crohn’s disease and ulcerative colitis, for which the response of patients to drugs is often difficult to quantify. Figures 4b and 4c show quantitative DHM phase images from investigating unstained cryostat colonic sections (thickness: 7 μm) of an animal study on healthy and colitic mice with inflamed intestine caused by dextran sodium sulphate (DSS). Corresponding hematoxylin and eosin (H&E)-stained samples are depicted in Figures 4a and 4d. The QPI images show the various anatomic tissue structures (epithelium, submucosa, and stroma), which are also visible in the H&E-stained sections. However, the retrieved quantitative DHM phase images (Figures 4b and 4c) can also be evaluated further to retrieve a refractive index map of the investigated tissue9. Bright regions in DHM phase contrast images indicate a high refractive index, which corresponds to increased tissue density (Figures 4a and 4d). In comparison, Figure 4e shows the average refractive indices for epithelium, submucosa, and stroma for healthy and inflamed tissue. For all layers of the inflamed colon, a significant refractive index decrease is shown. The detected refractive index decrease correlates with clinical parameters such as the relative loss of body weight of the investigated animals9.
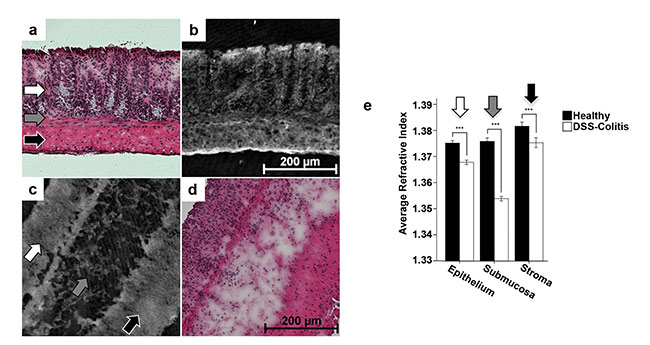
Figure 4. Quantitative DHM phase images of unstained cryostat colonic sections of healthy and colitic C57Bl/6 WT mice with inflamed colon (b, c). Corresponding H&E-stained samples (a, d). Analysis of the refractive index in various tissue layers (white arrow: epithelium; gray arrow:
submucosa; black arrow: stroma) revealed a significant tissue density decrease of inflamed tissue areas (e). ***Statistical significance. Adapted with permission from Reference 9.
The examples cited above illustrate the capabilities of DHM and related QPI techniques. The methodology quantifies morphology, growth, and mass of adherent and suspended single cells by biophysical parameters without labeling. This information can be used to distinguish among various cell types, quantify cell culture conditions, and obtain cellular response to toxins or drugs. In histology, QPI of dissected samples provides a stain-independent contrast and tissue density. QPI with DHM has the potential to be used as a powerful routine tool in various areas of biomedicine and toxicity testing, such as in cell biology, cancer research, nanotoxicology, and histopathology. The main challenges to future developments of the technology are the improvement of multimodal imaging and the simplification and acceleration of the procedures for image acquisition and data extraction. The Biomedical Center of the University of Münster and other groups of the international QPI community are therefore performing research on highly automated systems, suitable standard operations procedures (SOPs), and the integration of machine learning algorithms to achieve statistically convincing amounts of measurement data with minimized effort and time.
Meet the author
Björn Kemper, Ph.D., is a senior researcher at the Biomedical Technology Center of the Medical Faculty at the University of Münster, Germany. His research interests are optical metrology and biomedical optics, with a special focus on digital holography and quantitative optical microscopy for label-free imaging of biological cells and tissues. Kemper has developed various DHM systems and methods, and he has demonstrated their application capabilities for QPI in multiple application fields.
References
1. Y.K. Park et al. (2018). Quantitative phase imaging in biomedicine. Nat Photon,
Vol. 12, pp. 578-589.
2. B. Kemper and G. von Bally (2008). Digital holographic microscopy for live cell applications and technical inspection. Appl Opt, Vol. 47, pp. A52-A61.
3. B. Kemper et al. (2019). Label-free quantitative in-vitro live cell imaging with digital holographic microscopy. In Bioanalytical Reviews. Vol. 2. J. Wegener, ed. Springer Nature Publishing: Basel, Switzerland.
4. P. Lenz et al. (2016). Multimodal quantitative phase imaging with digital holographic microscopy accurately assesses intestinal inflammation and epithelial wound healing. J Visualized Exp, Issue 115, e54460.
5. P. Langehanenberg et al. (2011). Autofocussing in digital holographic microscopy.
3D Res, Vol. 2, Issue 4.
6. D. Bettenworth et al. (2018). Quantitative phase microscopy for evaluation of intestinal inflammation and wound healing utilizing label-free biophysical markers. Histol Histopathol, Vol. 33, Issue 5, pp. 417-432.
7. L. Kastl et al. (2017). Quantitative phase
imaging for cell culture quality control. Cytom A, Vol. 91, Issue 5, pp. 470-481.
8. L. Kunsmann et al. (2015). Virulence from vesicles: novel mechanisms of host cell injury by Escherichia coli O104:H4 outbreak strain. Sci Rep, Vol. 5, Article 13252.
9. P. Lenz et al. (2013). Digital holographic microscopy quantifies the degree of inflammation in experimental colitis. Integr Biol, Vol. 5, Issue 3, pp. 624-630.