
FRET-based drug screen detects kinases
David Shenkenberg
Kinases switch cellular pathways on and off by phosphorylating cellular
components. They are ideal drug targets because drugs that target them can control
entire pathways. Although Förster resonance energy transfer (FRET) can easily
monitor kinase activity in single cells, it cannot easily measure entire compound
libraries. Researchers have solved that problem by developing a FRET-based high-throughput
drug screen that detects the activity of protein kinase A (PKA) and cyclic adenosine
monophosphate (cAMP), the latter of which activates PKA.
Scientists in Jin Zhang’s laboratory at
Johns Hopkins University School of Medicine in Baltimore engineered new FRET-based
reporters by integrating modified versions of yellow and cyan fluorescent proteins
with proteins that interact with PKA and cAMP.
Zhang said that they chose FRET because
it is noninvasive, sensitive and provides spatiotemporal resolution. She added that
they could genetically encode fluorescent proteins in the cells used in their screen,
which they could not do easily with fluorescent dyes. However, dyes tend to emit
light more intensely than fluorescent proteins and have a greater dynamic range,
so the researchers circularly mutated the Venus domains of the proteins to make
them brighter and to increase their signal amplitude.
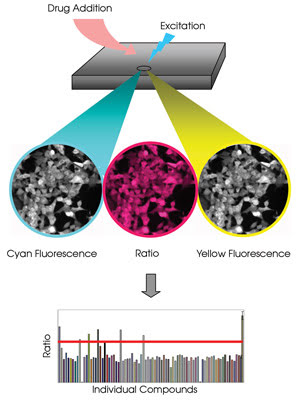
This FRET-based drug screen monitors kinase activity. Each well of the plate contains
living mammalian cells expressing a kinase biosensor. A plate reader measures cyan
and yellow fluorescence, and a computer calculates the emission ratios, resulting
in a graph showing responses generated by individual compounds.
When performing FRET assays with the
reporters, the fluorescence measurements lasted 64 to 92 seconds. Zhang said that
her group is attempting to hasten the process by devising a method to simultaneously
detect both fluorophores.
The researchers next modified the PKA
indicator with a nuclear export signal because kinase activities often are compartmentalized.
They saw an expected difference in kinetics due to the exclusively cytosolic location
of the reporter. Zhang said that they have developed a version of the reporter with
a nuclear localization signal, and they are currently testing it.
They found that the PKA indicator exhibited
high sensitivity and low variability, but that the cAMP reporter initially showed
only acceptable levels of sensitivity and variability. However, they improved results
for the latter indicator by stably transfecting the cells with the construct.
They validated their assay on a 160-drug
library of FDA-approved drugs and clinically relevant compounds, including lidocaine,
coumarin, aspirin and testosterone. All known agonists and antagonists produced
positive hits, and most drugs showed no effect. The researchers said that these
results prove the accuracy of their screen.
However, a few drugs confounded the
results because of their toxicity or colorimetric properties. In addition, bilirubin
unexpectedly inhibited cAMP. Although the researchers added a high concentration
of bilirubin, they suggested that their results could indicate a new mechanism of
bilirubin activity.
Before looking at interesting hits
individually, the researchers plan on continuing to test the rest of the compounds
in the library, which number more than 3000.
ACS Chemical Biology, July 26, 2006, pp. 371-376.
Published: September 2006