Designing a low-cost biomedical system isn’t easy. After all, it has to achieve acceptable repeatability and sensitivity while holding down expenses and meeting regulatory requirements.
That can be difficult, and some obvious avenues can be the wrong ones to follow. For instance, cutting component costs can be attractive, but skimping on the wrong thing can actually end up being counterproductive.
“Ordering parts strictly based on cost sometimes is going to cost you a lot more than you might save on the hardware,” said Matt Slaughter, machine vision business development manager for integrator Cyth Systems LLC of San Diego.
A look at some examples shows how biophotonic technology is helping to meet low-cost system design challenges. It also shows where to save and where to spend.
Cutting the cost of sequencing
The first example comes from the world of gene sequencing, where automated machines analyze light signals arising from fluorophores selectively attached to the nucleotides that are the code of life. Harvard researcher Dr. George Church wanted a system inexpensive enough for someone with a modest budget or grant to be able to do the sequencing.
Shown are four-color fluorescent beads, which are converted to base calls and used for genetic analysis in a sequencer designed to be low cost. Images courtesy of Dover.
Some years ago, Church’s team produced a prototype. They then worked with Dover of Westborough, Mass., to improve the system while cutting costs.
Dover’s chief technology officer, Kevin McCarthy, helped in this effort and characterized the resulting product, the Polonator G.007, by saying, “Basically it’s a very sophisticated, automated fluorescence microscope targeted at low-cost genome sequencing that can be used for a wide variety of applications.”
The system is about one-third the cost of other second-generation sequencers, McCarthy added, because of a number of cost-effective design choices. For example, it needed a way to interrupt a bright light source within a few milliseconds. Instead of a more expensive approach, this function was accomplished by mounting a vane on a $20 stepping motor, which twitches to block and unblock the light. This setup also allows the vane to act as an attenuator, thanks in part to a liquid lightguide.

Although designed to be low cost, the Polonator, a second-generation gene sequencer, doesn’t skimp on the image sensor, acquiring 2180 images per lane (right) in each of four colors for every base read. Each image is 320 µm2.
The filter indexing mechanism, which contains a filter cube for each of the four DNA bases, is mounted on the shaft of an inexpensive stepping motor. The result is fast switching between filter cubes and a positioning repeatability of less than 15 µrad.
The movement of the stage in X, Y and Z is handled by a custom linear motor based on inexpensive bent sheet steel and neodymium boron magnets. The positioning resolution is 5 nm, achieved at low cost by using a 12-bit analog-to-digital converter and a software-based interpolation of a signal from an analog encoder.
The cost of the filters in the system was reduced by mounting them in injection-molded plastic instead of putting them in machined metal. The filters were from Semrock Inc. of Rochester, N.Y., and offered what McCarthy characterized as very good performance at an inexpensive price.
One key to cutting the cost of the Polonator is that, although it can be thought of as an automated bright-field microscope with fluidics and temperature control, it is not like the typical scientific microscope. The optical path is a straight shot, so there is no need for folding mirrors that have to be aligned or adjusted.
All that is needed is a high-numerical-aperture objective, a tube lens and a camera, McCarthy said. “We simply buy high-end optical and imaging components – objectives, tube lenses and cameras – and precisely hard-mount them in place.”
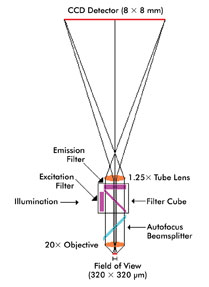
A straightforward optical path and a minimum of precisely mounted optical components – objective, tube lens and camera – help hold down the cost of a second-generation gene sequencer.
The camera is a high-end, electron-multiplying CCD. The choice was largely dictated by the need to detect photons efficiently while achieving the highest possible throughput.
Cutting the cost of the Polonator and follow-on systems should further lower the cost of sequencing an individual genome. The Human Genome Project that ended in 2003 cost billions. It took $70 million to sequence the second human genome, which was completed in 2007. McCarthy estimates that the Polonator today can do the job in a month for about $40,000.
The design and development of a lower-cost, higher-performance version is actively being worked on, he said. “We are pursuing a variety of additional advances in the system, and our ultimate goal is the sub-$1000 genome.”
Try before you buy
For another example of low-cost system design, consider lessons learned from solutions put together by Cyth Systems. The company makes starter systems outfitted with basic equipment and software. For these and other products, it offers support that can be critical to a successful final outcome, Cyth’s Slaughter said.
“Typically, we’ll allocate 10 hours of preapplication support. That’s us bringing your part into our lab and getting the right lighting and the right optics.”
Slaughter noted that life sciences requirements can be used to design a lower-cost system. For one thing, the application may be in a lab and not on a plant floor. Not having to deal with dust, vibration and water means that the system can be less impervious to the environment and, therefore, cheaper.
There may be no need for high-speed imaging or real-time control, both of which may require a costly real-time operating system or specialized hardware. Instead, a standard PC and its inexpensive software may be sufficient.
As for the camera, getting that right while holding down cost can be a bit trickier. Color data often is important in monitoring a dye or chemical process, so the camera must capture that. The typical color camera uses a Bayer pattern, which has twice as many green pixels as red or blue and approximates the color at each pixel.
Applications demanding true color data at all pixels may get better results from more expensive solutions. One possibility is to use a monochrome camera, appropriate filters and a mechanism to switch between them. Another solution is a three-sensor camera equipped with an internal beamsplitter that sends light to each sensor.
Sometimes, however, a low-cost USB camera may be all that’s needed. Slaughter cautioned, though, that this approach doesn’t necessarily mean using a consumer-grade camera.
For one thing, such cameras may have dead pixels in them, with their output simulated by averaging that of nearby pixels. The dead pixels may be hidden but still affect data collection. For another, consumer goods tend to have a short product life cycle.
Industrial-grade cameras, on the other hand, tend to be around longer. Also, if they have any dead pixels, they are not hidden. Finally, an industrial-grade camera may have an IP, or International Protection, rating. This may make an IP-rated enclosure unnecessary, and the savings may more than cover the additional cost of the camera.
Indeed, according to Slaughter, a design choice that reduces the number of components or the complexity of integration can pay for itself. Even if the components cost more, there may be enough labor saved to make up the difference.
A careful accounting of the total expense in parts and labor when designing a low-cost system is vital, he said. “The more planning you do for that up front, in the long term you have a much better idea of what your actual end price is going to be.”