Dr. Fran Adar, Jobin Yvon
Any given material can be characterized using a wide variety of spectroscopic techniques, each of which provides useful information. Depending on the material in question, of course, some methods are more effective than others. For example, fluorescence spectroscopy tends to work best on biochemical samples because it is a nondestructive method. However, the clever researcher realizes that a wider range of information can be collected from a sample by combining several measurement techniques.
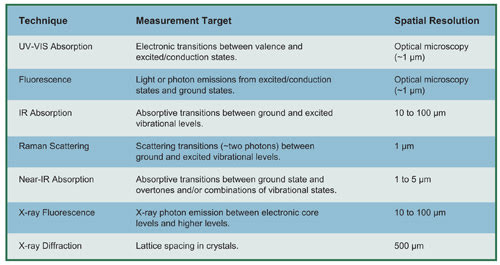
To evaluate the potential interest in multiple measurements, it is useful to first understand the spatial resolutions, sensitivities and physical properties of samples under analysis. The table below provides an overview of several spectroscopic methods.
Because Raman scattering and IR absorption both probe vibrational levels of the ground state, they have long been considered complementary techniques. Historically, analytical chemists using Fourier transform infrared (FTIR) spectroscopy felt that, because both methods probed the same physical states, there was no need to introduce Raman spectroscopy into the mix.
But the situation has changed drastically over the last 15 years because of the development and implementation of high-quality edge and notch filters, air-cooled lasers, low-noise multichannel detectors (especially CCDs) and Raman microscopes. As a result, the number of turnkey Raman instruments introduced into routine analytical laboratories has increased dramatically, and these instruments are beginning to see increasing use for solving real-world problems.
Anyone who has ever examined the FTIR and Raman spectra of the same products realizes how different they are, because the two techniques are sensitive to different characteristics. For instance, carbonyl functional groups often have very strong IR bands, but bands of medium intensity in the Raman. On the other hand, Raman spectra are quite sensitive to unsaturation (which can be useful in polymerization studies, for example).
One of the most important advantages of IR over Raman is the technique’s extensive spectral libraries, which can make problem solving painless. But Raman offers better access to lower energy bands. The low-frequency cutoff in Raman spectra is set by the cutoff of the laser filter; depending on the type of instrument being used, this can be anywhere between 5 and 150 cm–1. The low-frequency cutoff of FTIR instruments is determined more by the beamsplitters and detectors. (In FTIR systems, as the low-frequency cutoff is moved to lower energies, there is a subsequent increase in detection of blackbody radiation, which contributes to the noise in the spectra.) For most benchtop instruments, the low-energy cutoff is set between 450 and 650 cm–1.
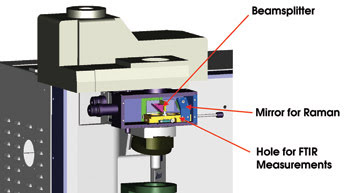
A switching device allows this instrument to alternate between IR and Raman modes. On the left, a beamsplitter injects white light into the sample, creating a white-light image together with the IR beam on the IR camera. In the middle is a hole for FTIR measurements. On the right, a mirror for Raman measurements enables standard Raman white-light visualization of the sample and of the laser spot simultaneously.
What does it all mean? If a researcher must detect many metal oxides, Raman is the method of choice. Included in this class of substances are hematite (Fe2O3), magnetite (Fe3O4), goethite and lepidocrocite (FeOOH), anatase and rutile (TiO2), lead oxides of many forms, and tetragonal, monoclinic and cubic ZrO2. Types of applications in which the detection of metal oxides is important include corrosion studies, and identification of pigments in paper, fibers, artwork and paints.
Modern analytical Raman and FTIR spectroscopies are used most frequently in the microscopic mode. Another microscopic method that can complement both of these techniques is micro x-ray fluorescence, which produces information on elemental composition. As with scanning electron microscopy, this technique can produce elemental maps, but sampling is not done in a vacuum chamber. Therefore, samples can be studied under routine ambient conditions without having to be specially prepared or treated. Most importantly, x-ray fluorescence can be employed to examine samples in water.
The biggest drawback to using this technique is its limited ability to focus the x-ray beams. However, thanks to new capillary-focusing elements, researchers can produce beam spots as small as 10 μm, making the spatial resolution similar to that of FTIR spectroscopy. Elemental maps can be quite helpful in combination with Raman maps when the sample examined is totally unknown.
Combining Raman and x-ray fluorescence techniques enables scientists to identify common metal oxides. Although one of the strengths of Raman spectroscopy is its unique ability to distinguish different physical states of the same chemical composition, interpreting the spectra of some inorganics without any elemental composition can sometimes be daunting because of the technique’s sensitivity to so many forms of material. But once the elements are known — and micro x-ray fluorescence can help collect that information — the number of possibilities becomes much more manageable. Scientists can then handily determine the composition of the phases that are present.
One field that is witnessing an increased use of spectroscopy for characterization is the pharmaceutical products sector. Here, both Raman and FTIR techniques are used to identify polymorph and solvate forms, which is useful for characterizing physical and chemical behaviors, as well as for patent protection (by enabling a drug company to patent a particular crystalline form, as well as the chemistry of the compound).
These two techniques also are commonly used to measure the distribution of the active pharmaceutical ingredient in final drug formulations. Again, however, these efforts can be complemented with x-ray fluoresence. For example, some excipients, such as magnesium stearate, are mixed with the final formulation, and a magnesium map could show investigators the distribution of those substances among the active pharmaceutical ingredient, microcrystalline cellulose, starch and lactose in the tablet.
Looking ahead, the combination of spectroscopic techniques will continue to pay off for investigators seeking to derive more information from samples. Already, industries benefiting from the combined power of hybrid techniques include analytical laboratories in manufacturing — such as pharmaceuticals, polymers and semiconductors — and corrosion engineering companies and art conservation groups.
Contact: Fran Adar, worldwide Raman application manager, Jobin Yvon, Edison, N.J., +1 (732) 494-8660; e-mail: [email protected].