Imaging is an expansive market where technological advances in microscopy, lasers and other techniques are resulting in a growing number of applications and functions.
Among imaging techniques, multiphoton excitation microscopy is becoming increasingly popular for biomedical research in areas such as neuroscience, where deeper and quicker imaging is needed. Fluorescence microscopy is a developing method for studying tissue in situ and in real time. Optical coherence tomography imaging is used in ophthalmology, in dermatology for diagnosis and treatment of skin cancers, and in conjunction with laser-based aesthetic treatments such as skin rejuvenation. Optical microscopes also are a leading imaging technology in life sciences research.

Imaging techniques, such as those used in microscopy, and tools including optical microscopes are becoming increasingly popular in biomedical R&D and the life sciences.
BioPhotonics spoke recently with several industry experts about the state of the global imaging market, as well as biomedical R&D and the life sciences, and the field’s potential. They are Jun Hee Kang, an application scientist at Spectrolight Inc., and a team from Coherent Inc. — Marco Arrigoni and Matthias Schulze, marketing directors for the scientific market and OEM components/instrumentation, respectively.
Q: What are the most notable imaging technology advances within the life science and medical fields, and what are the implications of such advancement?
Arrigoni: One of the most dynamic areas for imaging in life sciences is the use of multiphoton excitation (MPE) microscopy in neuroscience. Here, the overarching trends in technology advances are targeted at deeper and faster imaging in live tissue.
Specifically, MPE is used to study the structure and function of neural networks in the brain of model animals. What does this mean in terms of practical imaging capabilities? [Scientists] are beginning to investigate subcortical tissues. As other areas of the brain are studied … we expect to see a growing demand for even deeper imaging. At the same time, scientists are looking to simultaneously study ever-larger populations of neurons. By study, this usually means imaging action spikes using a calcium ion indicator. The timing and linking of these spikes is then observed following some type of stimulation of the animal or lately by targeted stimulation of selected neurons using optogenetics. There is a drive to eventually study thousands of neurons in a single column of mouse cortex under a MPE microscope.
The penetration depth of laser light in brain tissue is determined by absorption and scattering. Researchers have been increasingly using probes excited at longer wavelengths — 950 to 1200 nm — but in the last three years they started to work also in the two windows of maximum penetration — around 1300 nm and 1700 nm. As a result, we are seeing all kinds of advances in those wavelength regions.
Kang: The optical microscope is the dominant imaging mode in laboratory life sciences, and most of the technology advances continue to be in this field. Image acquisition speed continues to be increased across the field, in order to support the study of dynamic events such as neuron signaling in real time. Recent advances in light sources, cameras and scanning techniques all support faster imaging — either directly or by improving the signal/noise of images, and thereby shortening the necessary acquisition times. The demand for increased speed is also driven by the growing use of multidimensional data.
[In regard to] the use of multiple wavelengths … simply stated, the optical microscope has a unique ability to map samples for composition in addition to structure. But this is all predicated on wavelength discrimination. Scientists use lasers or filtered lamps to preferentially excite fluorescence in a dye, or probe an expressed protein (referred to as a fluorophore) that is attached to structures or molecules of interest. A filter in front of a CCD or CMOS camera then typically blocks the scattered laser light so that only the Stokes-shifted fluorescence is efficiently detected. Today’s experiments often involve mapping multiple fluorophores, each with different wavelength properties. The most comprehensive data set includes XY images at multiple Z planes in the sample, and at multiple wavelengths, all followed in real time.
The ultimate wavelength-filtering device is the monochromator. You can independently scan the center wavelength and adjust the bandwidth of transmission. But because it disperses wavelength as a function of displacement in the exit plane, this leaves only one spatial axis available for imaging. Indeed, you virtually never see a monochromator hooked up to a camera in a microscope setting. Instead, scientists use one or more bandpass filters, often in the form of a filter wheel. These filters have fixed properties that limit the kind of experiments you can do.
Q: How do you perceive the state of the imaging market as it relates to the life sciences?
Kang: Imaging is obviously a very broad term, even just within the biomedical sphere. Optical imaging techniques dominate the research field in life sciences, where most of the focus is on microscopic scale imaging. Similarly in clinical laboratory work, the optical microscope is a pre-eminent tool for tasks such as tissue biopsy. The majority of applications in both cases rely on some type of fluorescence microscopy.
But then when you look at medical imaging, optical techniques are just a tiny fraction of the total. There have been some notable successes in studying tissue in situ — real-time biopsy — again based on fluorescence microscopy. Plus, OCT is undoubtedly a growing technique. But in both cases we are talking about tiny niches of the overall medical market. So in terms of interesting developments in imaging, most of the photonics action continues to be in the research field and centered around fluorescence microscopy.
Schulze: A large proportion of imaging applications in life science research applications rely on the detection of fluorescence, which supports very high signal-to-noise, allowing detection down to single molecule levels. In spite of advances in LED technology, the laser is still widely used as the light source of choice because of its unique advantages such as spectral brightness (monchromaticity) and spatial brightness (focusability), plus the femtosecond temporal resolution afforded by fast-pulsed lasers.
In methods based on CW lasers, a notable advance in recent years has been the development of superresolution methods that finally breached the diffraction limit. These methods produce incredibly detailed images that are enabling scientists to study biological structures and processes as never before. The optically pumped semiconductor laser (OPSL) [was] the first solid-state laser technology that was scalable in terms of power and wavelength. Today, the OPSL dominates life science applications at 488 nm. Just as importantly, OPSLs have been developed at other wavelengths, enabling confocal microscopy to move beyond the limits of a single laser wavelength with the corresponding development of fluorophores at numerous new wavelengths.
Imaging technology advances are enhancing the study of biological systems such as neurons.
Applications for laser-based fluorescence microscopy are incredibly diverse throughout life sciences research. Particularly active areas are neuroscience, as well as in gene control/expression, especially where this relates to pharmacology. New laser wavelengths continue to be developed to match the needs of newly developed fluorescent probes.
Q: What is the potential for imaging technology in life sciences and medical applications?
Kang: Optical imaging methods such as microscopy enable direct visualization of structure. But when you add in wavelength selectivity, you can map content. Optical imaging is virtually unique in this noncontact interrogation capability. That’s why even as the frontiers in life sciences are advancing faster than ever before, the venerable and humble microscope is still a frontline tool. Looking to the future, in the research field and also in clinical labs, observations with an optical microscope will continue to yield Nobel prizes and provide new treatments for cancer and other diseases.
Medicine is another matter. Until now, outside of the pathology lab, the physician’s eyes have been the only widely used optical imaging tool. But I think the Vein Probe [a device that shows high-resolution images of blood vessels in real time using IR detection] is possibly just the first of many advances in the use of photonic-based tools for macroscopic imaging in medicine.
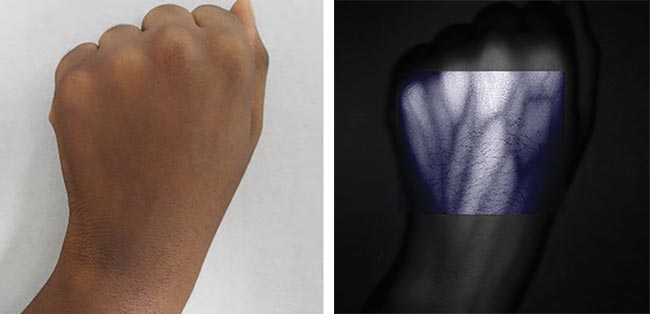
A Vein Probe — an imaging device that shows high-resolution images of blood vessels in real time using IR detection — enables improved imaging and visualization of vascular details in human and veterinary patients. Courtesy of Spectrolight Inc.
I think it’s safe to forecast that there will be many more advances and changes; we live in very dynamic times in the photonics industry. In terms of specific methods and technologies, it is difficult to guess exactly where the next clever idea is going to be. However, I think the exciting synergism between end users and technology developers will continue forever, with the same wonderful “chicken and egg” scenario as today. By this I mean that on the one hand, if researchers clamor loudly for a capability, clever engineers at a photonics company will come up with a solution. And conversely, whenever a company comes up with a new device … end users quickly dream up new applications and experiments that are newly enabled by this device.