Years after Aydogan Ozcan’s demonstration, the technology evolves to meet real-world applications.
HANK HOGAN, CONTRIBUTING EDITOR
With advancements in image processing and sensors, lens-free microscopy is ready for a closer look by researchers, industry, and consumers. Compared to traditional benchtop microscopes, lens-free techniques can cost less, reduce instrument size, enlarge the field of view, and improve throughput.
“The device [a lens-free microscope] is 10× smaller than a regular microscope. But the image, on the contrary, is 100× larger than any [other] microscope image,” said Cédric Allier, a project manager with the Grenoble, France-based research organization CEA-Leti.

Aydogan Ozcan displays hand-held, lens-free microscopes. The black one (left) is installed on a phone and uses the phone’s CMOS imager for lens-free microscopy of a specimen. The white prototype (right) is a stand-alone hand-held lens-free microscope that is controlled by a laptop using a USB cable. This stand-alone version weighs less than 50 g. Courtesy of Ozcan Lab/UCLA.
The large field and depth of view makes it possible to look at perhaps 10,000 cells at once. Therefore, the lens-free approach is well suited to needle-in-a-haystack problems, such as detecting cancer or other diseases. The small size, low cost, and ruggedness of lens-free microscopes also make them useful for hand-held and portable devices that are capable of detecting medical problems or performing environmental monitoring in the field.
Several small companies and startups are either actively producing lens-free products or plan to do so soon. If lens-free methods offer the cost, performance, and usability necessary to achieve commercial success, these products promise to boost public health by identifying waterborne toxins, airborne pollutants, and infectious diseases in the field.
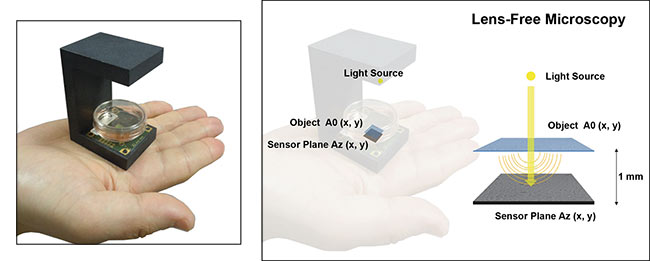
A lens-free microscope consists of a semicoherent light source illuminating an object that sits directly atop an image sensor, a design that gives rise to holograms
that can be reconstructed into images. Courtesy of CEA-Leti.
How technology has changed
In a conventional microscope, light interacting with a target is collected and focused by lenses. Lens-free imaging, as the name implies, eliminates such bulk optics. Instead, based on a concept that first appeared in papers co-authored by Aydogan Ozcan, a UCLA electrical engineering and bioengineering professor, these on-chip microscopes have a light source located centimeters from a specimen that sits directly atop a CMOS image sensor.
The light is partially coherent, giving rise to an interference pattern captured by the sensor. Processing this holographic data using an appropriate algorithm designed specifically for the task creates an image suitable for viewing and analysis. The software also makes it possible to focus digitally rather than mechanically.
A key to making this setup work is the image sensor, according to Allier, who has been working on lens-free techniques for a decade. He said the pixel size of the sensor helps determine the resolution of the microscope. In 2010, readily available image sensors had a roughly 5-µm pixel size. By 2015, this had dropped to 1.7 µm, making it possible to image cells. The pixel size continues to shrink and is now as small as 0.8 µm.
What’s more, over the last few years, increasingly powerful GPUs, or graphics processing units, have shortened the image processing time. A half decade ago it may have taken an hour to do this processing for a 2D image, Allier recalled.
“Today it takes less than 30 seconds, thanks to the GPU and the fancy algorithm we’ve developed,” he said. This speed makes 2D lens-free microscopy acceptable for clinical work.
These microscopes use a monochromatic, narrowband LED that shines through a 100-µm diameter pinhole, in CEA-Leti’s version. This source is coherent enough without the cost or power requirements of a laser.
IPRASENSE, a company based in Montpellier, France, was founded to commercialize the CEA-Leti technology, including the holographic reconstruction algorithms, acquisition setup, and software as well as the data analysis chain. The almost 7-year-old company has a line of cell-culture monitoring equipment that uses lens-free microscopy and automated algorithms to rapidly count cells and determine their viability.
The automated approach is about
10× faster than the manual equivalent, Allier said. Tests have shown the two methods provide comparable results, and the automated approach doesn’t require skilled personnel in its operation.
After their initial papers on lens-free imaging, Ozcan’s UCLA group produced a 2010 Optics Express paper describing much better resolution results than those set by pixel size. The investigators did this by shifting the aperture, and therefore the illumination center, in small steps across the object. They then combined the shifted holograms into the final image.
“This method computationally divides each pixel into smaller pixels to be able to reach the diffraction limit of light,” Ozcan said. He added that later work showed shifting the sensor achieved the same effect.
This pixel-segmenting technique does require the capturing and processing of more data, and a more complex microscope than otherwise would be the case. But in addition to offering higher resolution, the stepped approach also reduces speckle noise that arises from wavelength-scale object surface roughness and reduces unwanted reflection interference, improving image quality.
Marketplace applications
Ozcan’s group has explored a range of possible microscopy applications, including portable units that count and classify algae in seawater. These could provide a faster and more field-friendly means to detect algal blooms, which would be a public health benefit, as such overgrowths can create toxic conditions that shutter beaches.
The technology could be used to determine the presence of airborne pollen, mold spores, and other allergens, information important to allergy sufferers. It could also be used to screen for parasites and other diseases, in the field and at the point of use.
To commercialize the technology, Ozcan spun off two companies, Cellmic LLC and Lucendi Inc. Both are based in Los Angeles.
“Cellmic has been focusing on point-of-care diagnostics and mobile health applications. Lucendi has been focusing on high-throughput liquid analysis, including environmental monitoring and water analysis,” Ozcan said.
Sales of Lucendi products should start in 2019, he added. In 2018, CEA-Leti licensed a core patent from Cellmic, and this technology is now working its way into more products and applications.

A lens-free microscope that performs pixel-superresolution holographic on-chip lens-free microscopy in a hand-held design. It can image phase and amplitude information of transmissive samples (such as tissue samples, cells, pathogens, etc.) with a resolution that is better than the size of each pixel of the CMOS imaging chip. Courtesy Ozcan Lab/UCLA.
The arrival of open-source machine learning platforms such as TensorFlow is another recent innovation that is advancing these technologies and their applications, Ozcan said. With machine learning, software is presented with the raw data and the desired outcome in example cases where the final image is known. It then figures out how to transform the data into the image.
As a result, physicists and engineers have ways to reconstruct lens-free holograms more efficiently and rapidly than before. Machine learning techniques recently led to lens-free holographic imaging with the color and contrast of a light-field microscope, something not possible previously, Ozcan said.
He noted that lens-free microscopes can be made for under $50, excluding the computer used for processing. The device weight can be less than 50 g, with a size small enough to fit in the hand. Importantly, there are no lenses to align and no focus to adjust, making the microscope more useful in field applications.
A focus on the future
In addition to the long-time lens-free microscopy researchers, there are some relative newcomers to the field. For example, Euan McLeod is now an assistant professor of optical sciences at the University of Arizona in Tucson who worked in Ozcan’s group at UCLA. McLeod is investigating lens-free microscopy applications that push the boundaries of sensitivity and seek to detect very small objects, such as viruses or individual protein molecules.
This might be done, he said, via functionalized beads. Coating these microscopic particles with biomarkers that visibly react to a specific microorganism, virus protein, or DNA strand makes it possible to image tiny targets. This approach could be used to observe and count biomarkers for detection of diseases, including cancer.
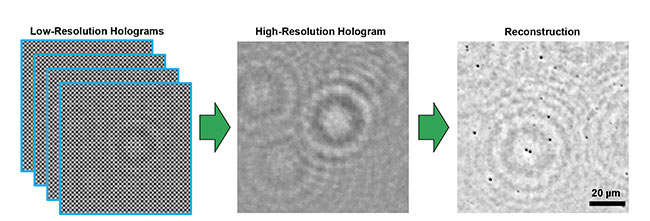
In lens-free imaging, computational algorithms combine low-resolution holographic data (left) to create a high-resolution hologram (center), which then
undergoes reconstruction into a microscopic image (right). Courtesy of Euan McLeod/University of Arizona.
Screening tissue slices for cancer requires viewing a very large number of cells at subcellular resolution. “You need a very large field of view and at the same time good resolution, with ‘good’ meaning micron or submicron resolution,” McLeod said.
Another recent entrant into lens-free microscopy is imec, the Leuven,
Belgium-based research organization.
At the 2017 SPIE BiOS conference in
San Francisco, imec investigators reported on the development of a lens-free holographic microscope built using a 1.12-µm-pixel sensor that achieved micron-resolution imaging. However, citing ongoing confidential research and active work in this area, an imec spokesperson said no further details or updates were available.
The University of Arizona’s McLeod noted that lens-free methods rival the resolution possible with traditional benchtop microscopes. In the past, light-induced artifacts were an issue. However, he said these resolution-robbing effects can be eliminated by shifting the light source location or its wavelength and capturing multiple images. Software can then combine the data and remove artifacts, thereby improving image clarity and usefulness.
McLeod noted that some imaging methods present challenges to lens-free microscopy. Fluorescence-based techniques, for instance, can be problematic because light emitted by the specimen is incoherent with the illumination, thereby destroying the interference that forms the basis for holographic imaging.

A prototype of a 3D lens-free video microscope for the monitoring of 3D cell culture. The specimen sits atop the image sensor chip (left).
A 3D reconstructed volume (4.7 mm3) of a 3D cell culture of prostatic cells (right). Courtesy of CEA-Leti.
Another issue that needs to be further addressed is imaging speed. According to CEA-Leti’s Allier, lens-free 3D imaging today is too slow for anything but demonstrations. It takes an hour, for example, to complete the image processing of a mosquito wing or other objects of about 5 cubic millimeters volume. In the future, faster GPUs should speed up this process. Machine learning could also lead to the development of new, more efficient, and faster image reconstruction algorithms.
There are several ways in which the continuing evolution of lens-free microscopy could have an impact, particularly in applications where the new technology offers an advantage over traditional forms of microscopy. On the clinical side, lens-free methods may make it easier to detect health problems, particularly infectious diseases in the field. The new microscopes, with their ability to rapidly and efficiently screen cells, could also benefit research and drug development.
On the consumer side, lens-free microscopy could lead to hand-held devices that enable people to measure and monitor air quality by imaging airborne particles, dust, mold, and allergens. The microscopes could similarly monitor water-quality issues. There have already been demonstrations of such possibilities. For example, Ozcan’s group reported, in a 2010 Lab on a Chip paper, on a cellphone attachment that operates using this technology.
According to Allier, further developments that improve device performance, such as speeding up lens-free 3D imaging, could spur new uses for lens-free microscopy.
“This will open up new applications that we haven’t thought of,” he said.