Jan Provoost, Richard Stahl, IMEC
A compact lens-free microscope could replace microscopes – and even expand their use – in a number of key biomedical applications, such as stem-cell culturing or in vitro drug testing. Using photonic integration, the microscope can even be microsized and used in advanced lab-on-chip systems.
More than half a century ago, Hungarian electrical engineer and physicist Dennis Gabor had the idea to add depth information to photos by capturing the interference between an illuminating wavefront and the diffraction of an object with a photographic plate. This marked the birth of holography and holographic imaging and earned Gabor the 1971 Nobel Prize for physics.
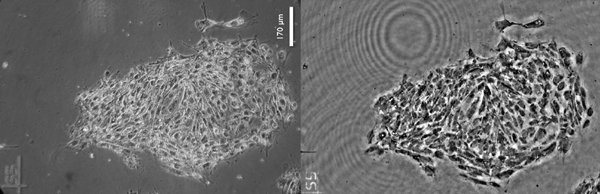
Stem cells seen through a phase-contrast microscope (left) and through imec’s lens-free microscope (right).
Today, advances in complementary metal oxide semiconductor (CMOS) technology allow the photographic plate to be replaced with a fast, high-resolution, digital image sensor. Computers have become powerful enough to reconstruct and process the image within a reasonable time frame, so it has become practical to convert the captured holograms into useful images by calculation instead of by illumination.
The concept has led imec to develop a lens-free microscope: an industrial-strength solution (not a one-off lab demonstrator) that can be mass-produced and integrated with biotech and life sciences tools. This lends the possibility of inspecting living transparent cells, monitoring cultures and tissues using the large field of view, and automating cell identification and inspection with smart image processing. Prototypes were validated and optimized for specific biotech applications: automated stem-cell culturing, drug screening on heart cell cultures and automated blood cell identification.

A prototype of imec’s lens-free microscope. Courtesy of imec.
Visual monitoring of stem cells
The principle of a lens-free microscope using inline holography is simple: Take a coherent light source (e.g., a laser) and shine it through the transparent sample. On a digital image sensor, record the interference between the light that has passed through the sample and a reference beam. Then use this digital hologram to visualize the sample and extract information. This setup requires only a light source, an image sensor and a powerful processor to compute the images in (or near) real time.
To inspect a colony of stem cells, a microscope should be able to see individual cells but also should allow an overview of the dense colonies. This requires an imaging resolution of at least 1.3 μm, comparable to phase-contrast microscopes, but combined with a much larger viewing field of a few tens of square millimeters.
In traditional microscopes, the resolution is inversely related to the size of the field of view. That is, at higher resolution, the focus is on a smaller detail. The only way to increase the field of view is to move the object mechanically under the microscope, take many pictures and stitch them together. This requires a complicated, expensive setup and also disturbs the cell cultures though vibrations and temperature changes.
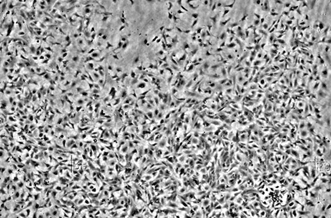
A cardiac cell culture seen through the lens-free microscope (horizontal view approx. 2 mm).
By contrast, in a lens-free setup, the field of view depends only on the size of the image sensor. For our prototype microscope, we selected a state-of-the-art, monochrome 10-MP imager with a 1.67-μm pixel pitch and a fiber optic light source. With this setup, we obtained a field of view of more than 29 mm2.
Arriving at an imaging resolution of 1.3 μm or better proved more challenging, mainly because of a side effect of our imaging technique, called twin image distortion. The solution is to recombine iteratively a number of slightly different input images, a technique known as iterative phase retrieval. An innovative approach is to use a series of images, each illuminated with a slightly different wavelength. Introducing a multiwavelength light source results in a robust, lens-free system with a 1.23-µm resolution, similar to the resolution of a phase contrast microscope with 10× objective, but with a field of view that is 14 times larger.
Stem-cell incubators
Until now, culturing stem cells has been a labor-intensive process and has been prone to failure. Stem cells grown in incubators have to be inspected regularly by laboratory assistants using microscopes to see whether the cells are thriving and when they can be harvested. Even the tiniest change in the environment of the cell culture – a change in light, temperature or vibration – may cause the colony to wither. With a field of view of 29 mm2, it is possible to monitor a cell colony and its individual cells at the same time. First, however, the colony must be located and brought into focus.
Cell culturing is often done on petri dishes with diameters of 30 or 60 mm.
To locate the colony, an automated microscope needs either a positioning system or a field of view that can scale to the size of the culturing media. Both are possible with the new lens-free setup. For tracking, a two-step positioning and acquisition approach is effective. This starts with a high-speed, low-quality image of a full petri dish, which enables location of the colony. A second step involves multiwavelength image acquisition and reconstruction for maximum quality.
Alternatively, it also is possible to create an ultralarge field of view by placing image sensors next to each other in a near-stitchless setup, which is not possible with systems using lenses. As for focusing, this system can compute the focus plane from the image – a digital autofocus. All depth information is available in the captured diffraction patterns.
This compact solution is ready to be integrated in an incubator environment. Mechanically robust, with no system recalibration or realignment necessary, it requires no maintenance. It can easily be scaled to a multimicroscope system with an ultralarge field of view. This is crucial to upscale stem cell production, lowering its cost and enabling more therapeutic applications.
Cell monitoring for drug discovery
A trend today in drug discovery is to test potential new drugs on lab-grown cells and tissues. To monitor contracting heart cells and to follow the conduction speed from one cell to the next in a monolayer of cells, researchers need to observe the rapidly changing shape of a relevant number of cells. This application requires a large field of view and a high imaging speed.
The speed depends not only on the speed of the image sensor but also on the speed with which the pictures can be digitally computed, either in real time or with postprocessing. For high-speed setups, the lens-free system’s computation and hardware setup had to be optimized using a combination of CPU and GPUs. The use of postprocessing resulted in an imaging speed of almost 300 frames per second, with higher speeds possible.
Heart cells contract when they receive a sufficient electrical stimulus from a neighboring cell, but drugs can interfere with that reaction. The next challenge was to measure the relation between the contraction of a heart cell and the electrical stimulus. To do that, imec researchers grew the cells on a chip with an array of microelectrodes. These surfaces are not transparent, so it is not possible to shine light through a sample and capture the result at the other side, as was possible in the first lens-free setups.
The new setup required a second innovation: reflective holography, where the hologram is made from the interference of the reference beam with the light that reflects off a sample (instead of the light that shines through the sample).
Integrated photonics: microsizing microscopy
A relatively small microscope can be used to study cell cultures growing in a petri dish, and there is no need to make the microscope microscopic. However, in a lab-on-a-chip and next to the microfluidic channels and chambers for chemical reactions, the microscope must be small, cheap and without mechanically moving parts. If all components can be microsized, a lens-free system is ideal.
For obvious reasons, a large field of view is not so important here. However, the resolution is important because the microscope in the lab-on-a-chip looks at cells, bacteria and large molecules. The resolution is determined by the image sensor’s pixel size. These are constantly getting smaller but are still in the range of 1.5 to 2 µm. To overcome this, a microscopic pinhole can be placed between the light source and the sample, magnifying the diffraction pattern onto the sensor. By varying the distance between the pinhole and the sample, the magnification can be varied without using lenses.

This prototype of the cell-sorting chip incorporates the lens-free system.
With such a system, we managed to identify three classes of cells from a blood sample (granulocytes, lymphocytes and monocytes) with an accuracy of 93%.
As a next challenge, this cell-sorting system can be scaled up to 1000 channels, each inspecting 1000 cells per second to determine whether they are malignant and mechanically sorting them into separate chambers. Each channel will have a lens-free microscope that will record a hologram of a cell, reconstruct and analyze the image, and then make a decision in a millisecond. The solution will use integrated photonics to distribute a single light source over the separate channels.
Exciting prospects
A number of biomedical applications could benefit from this lens-free system. Depending on the application, the prototype system is tuned toward multiple-cell, single-cell or subcellular resolution. Furthermore, fully automated, real-time monitoring could enable massively parallel and cost-effective drug screening.

A lens-free image of single-cell inspection (white blood cell).
These developments will transform lens-free microscopy from an R&D concept to a robust solution that can be fabricated by tool providers and used in commercial labs and industrial environments. Compared to classical microscopy, our technology is more robust, less complex to handle and has a larger field of view. It also offers the prospect of integration and scaling, as in an incubator environment and even a microsized lab-on-a-chip.
Meet the authors
Jan Provoost is science editor at imec in Leuven, Belgium; email: [email protected]. Richard Stahl is senior researcher for integrated imagers at imec; email: [email protected].