
Light-Activated Hybrid Bots Drive Muscle Activity Through Nerve Cells
Researchers from the University of Illinois at Urbana-Champaign have developed soft, microscopic, swimming biohybrid robots powered by skeletal muscle tissue that is stimulated by onboard motor neurons. The neurons have optogenetic properties — upon exposure to light, they fire to actuate the muscle tissue. The body of the biohybrid swimmer consists of a free-standing soft scaffold, skeletal muscle tissue, and an optogenetic stem cell-derived neural cluster containing the motor neurons.
The research team had previously developed self-propelled swimming biobots powered by beating cardiac muscle cells derived from rats. “That generation of singled-tailed bots utilized cardiac tissue that beats on its own, but they could not sense the environment or make any decisions,” professor Taher Saif said. In the new study, the researchers applied an optogenetic neuron cell culture, derived from mouse stem cells, adjacent to muscle tissue to create a biohybrid robot.
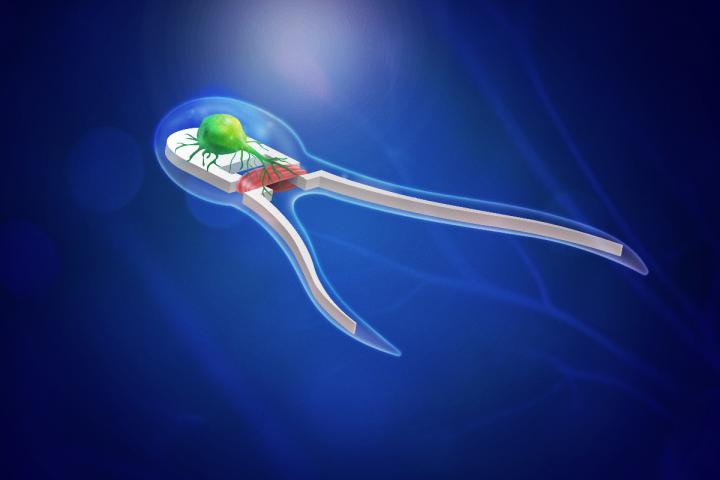
An artist rendering of a new generation of biobots — soft robotic devices powered by skeletal muscle tissue stimulated by onboard motor neurons. Courtesy of Michael Vincent.
Researchers built the biohybrid bots by embedding myoblasts in an extracellular matrix. When the myoblasts then self-organized into muscle tissue, guided by the geometry of the scaffold, scientists cocultured the muscle tissue in situ with a neural cluster. Motor neurons extended neurites selectively toward the muscle and innervated it, developing functional neuromuscular units. “The neurons advanced toward the muscle and formed neuromuscular junctions, and the swimmer assembled on its own,” Saif said. Cyclic muscle contractions, induced by neural stimulation, drove time-irreversible flagellar dynamics, providing thrust for the swimmer's untethered forward locomotion.
After the researchers confirmed the neuromuscular tissue was compatible with the synthetic biobot skeletons, they used computational models to optimize the swimmers’ abilities, determining the physical attributes, such as tail length, that would lead to the most efficient design.

The research team includes (from left) professor Taher Saif, graduate student Onur Aydin, graduate student Xiastian Zhang, professor Mattia Gazzola, graduate student Gelson J. Pagan-Diaz (seated) and professor and dean of the Grainger College of Engineering, Rashid Bashir. Courtesy of L. Brian Stauffer.
The researchers believe that this advancement could lead to the development of multicellular engineered living systems able to respond intelligently to environmental cues, with applications in bioengineering, medicine, and self-healing materials technologies. “Given our understanding of neural control in animals, it may be possible to move forward with biohybrid neuromuscular design by using a hierarchical organization of neural networks,” Saif said.
The team acknowledges that, like living organisms, no two biohybrid machines will develop to be exactly the same. “One may move faster or heal from damage differently from the other — a unique attribute of living machines,” Saif said.
The research was published in Proceedings of the National Academy of Sciences (https://doi.org/10.1073/pnas.1907051116).
Published: September 2019