A specially designed probe creates an optical response to the RNA sequences of SARS-CoV-2. The response is then measured by the sensor.
DUNCAN STACEY, LINKAM SCIENTIFIC INSTRUMENTS, AND XIAOGUANG WANG, THE OHIO STATE UNIVERSITY
The global outbreak of COVID-19 has prompted scientists worldwide to focus their research on the virus over the last year, particularly those who work with optical and sensing technologies. Some have concentrated on understanding fundamental aspects of the virus, such as its replication and transmission, while others have worked on creating vaccines or therapeutics. Many researchers and clinicians began to search for a reliable method to rapidly test a large proportion of the population to diagnose infected individuals as quickly as possible.
Recent groundbreaking work (Figure 1) at The Ohio State University1 (OSU) has led to the development of a rapid detection method for the SARS-CoV-2 virus using a liquid crystal sensor. The presence of the virus can be identified using visible light with this temperature-dependent sensor.

Figure 1. Adil M. Rather (left) and Yang Xu, members of professor Xiaoguang Wang’s group at The Ohio State University, work with the Linkam temperature control setup in their laboratory. Courtesy of professor Xiaoguang Wang/The Ohio State University.
Need for testing technology
As the pandemic spread, researchers rallied to develop testing protocols for what was then a largely unknown and potentially misunderstood virus. Various specialists turned their attention to SARS-CoV-2 testing and how they could adapt or pivot their research to tackle what was rapidly becoming a global threat.
Tests using reverse transcription polymerase chain reaction (RT-PCR) — the gold standard for molecular clinical diagnostics — became available quickly; however, RT-PCR tests require relatively long characterization time and specialized equipment to perform. A variety of other technologies have shown promise as candidate diagnostic tools — gold nanoparticles and column agglutination technology, for example — but each has significant limitations. The need was growing for fast, simple, and effective techniques to detect and identify the virus.
The team at OSU responded to the challenge. It is commonly known that thermotropic liquid crystals have been employed for detecting various biomolecules — for example, DNA and lipids. However, the team knew of no reports in which liquid crystals had been used to detect RNA, so it set out to apply the technique to SARS-CoV-2 RNA.
Since 2016, professor Xiaoguang (William) Wang has provided the research community with insights into the design of novel dynamic materials and systems based on colloidal and interfacial phenomena (Figure 2). This knowledge forms the basis of a novel class of stimuli-responsive materials that is used in a wide range of applications, such as pharmaceuticals and biosensors.
![Figure 2. A schematic shows the physical arrangement of the liquid sensor. The alignment before the addition of ssRNACoV (left). The change in alignment induced by the binding of the ssRNACoV target to the ssDNA probe (right). The image captures show the visual change from occluded (left) to clear (right) (insets). DMOAP: dimethyloctadecyl[3-(trimethoxysilyl) propyl]ammonium chloride. Courtesy of professor Xiaoguang Wang/The Ohio State University.](/images/Web/Articles/2021/3/23/Feat_LiquidSensors_Fig2.jpg)
Figure 2. A schematic shows the physical arrangement of the liquid sensor. The alignment before the addition of ssRNACoV (left). The change in alignment induced by the binding of the ssRNACoV target to the ssDNA probe (right). The image captures show the visual change from occluded (left) to clear (right)
(insets). DMOAP: dimethyloctadecyl[3-(trimethoxysilyl) propyl]ammonium chloride. Courtesy of professor Xiaoguang Wang/The Ohio State University.
Thermotropic liquid crystals
Liquid crystals exhibit predictable reactions as a result of their long-range orientational order and the mobility of their mesogenic constituents. They have been widely used in fast-switching electro-optical devices, such as liquid crystal displays. Liquid crystals are highly sensitive to mechano-optical changes or electric field variations, and the ability to monitor these changes by observing the liquid crystal orientation means that liquid crystals are increasingly used for sensing applications. Such applications include sensing electrical, mechanical, or vibrational changes, as well as identifying volatile organic compounds. Liquid crystals have potential use in a wide range of sensing applications due to their capacity for tunable, stimuli-responsive properties and relatively simple self-assembly and ordering characteristics.
The OSU team collaborated with professor Xiaoping Bao at Purdue University to design biosensors for SARS-CoV-2, and with professor Rongjun Qin at OSU to develop a smartphone app for automatic readout of the single-stranded RNA (ssRNA) of the virus.
Liquid crystals are ultrasensitive to small chemical modulations, and various analytes can be detected at extremely low concentrations. Therefore, taking advantage of the high sensitivity of liquid crystals, the researchers wondered whether they could develop a rapid detection method for the SARS-CoV-2 virus that would need no sophisticated equipment or infrastructure. So they set out to design a portable and economical liquid crystal-based sensor detection kit for rapidly detecting the ssRNA of the virus.
Over the past decade, a series of important works have revealed the design of liquid crystal films and droplets that undergo orientational ordering transitions in response to the adsorption of a wide range of molecules at an interface. For example, single-stranded DNA (ssDNA) and double-stranded DNA (dsDNA) produce different orientations of liquid crystals at cationic surfactant-laden aqueous-liquid crystal interfaces. This phenomenon leads to a change in the effect on visible light, caused by the optical birefringence of the liquid crystal film.
Proof of concept
To show that ssRNA from the virus exhibited the same property and that liquid crystals were suitable for COVID-19 detection, the OSU group followed a four-step experimental program.
First, a cationic surfactant dodecyltrimethylammonium bromide (DTAB)-decorated interface on micrometer-thick films of a commercially available nematic liquid crystal, E7, was prepared. The layers were supported on a dimethyloctadecyl[3-(trimethoxysilyl) propyl]ammonium chloride (DMOAP)-functionalized glass slide, which induced a perpendicular ordering of the E7 mixture.
Second, an ssDNA probe, with a sequence complementary to the defined SARS-CoV-2 ssRNA target, was adsorbed at the DTAB-decorated aqueous-E7 interface. The negatively charged ssDNA is attracted to the cationic DTAB at the aqueous-E7 interface via electrostatic interactions.
Third, ssRNACoV was added to the DTAB-laden E7 surface using the adsorbed ssDNA probe. The temperature of the system was increased to 48.7 °C, which is the melting temperature of the ssRNACoV. A Linkam PE120 Peltier hot stage was used to control the temperature of the E7 surface during the measurements. The ssRNACoV in melt form binds to the liquid crystal, inducing a change in orientation, which itself results in a visible change (from occluded to clear) that can be observed by using visible light microscopy via a simple smartphone app.
The researchers also used the PE120 to control and observe the temperature-
dependent phase transitions of liquid crystals, as well as to perform the polymerization of reactive liquid crystal mixtures at a specific temperature.
Finally, the optical appearance, or brightness, of the RNA-adsorbed E7 films was quantified from images captured by a CCD camera on a polarized microscope.
The team said these initial experiments showed that liquid crystal-based sensors offer a highly sensitive, reproducible, and robust method of detecting target ssRNACoV. The researchers also said they could establish the detection limit of the E7 surface for the target ssRNA by varying the concentration from nanomolar to femtomolar amounts.
Figure 2 illustrates this optical response.
Building a sensor
With the principle of sensor structure and detection performance established, the group moved on to prepare a final sensor design (Figure 3) that included using polarizing filter layers to enable visible light detection of the confirmation change in the liquid crystals. In addition, the sensor device was integrated with a retaining clip and an appropriate smartphone app to ensure consistent illumination of the sensor. This allows nonexpert users to reliably read and act upon the results of a test (Figure 4).
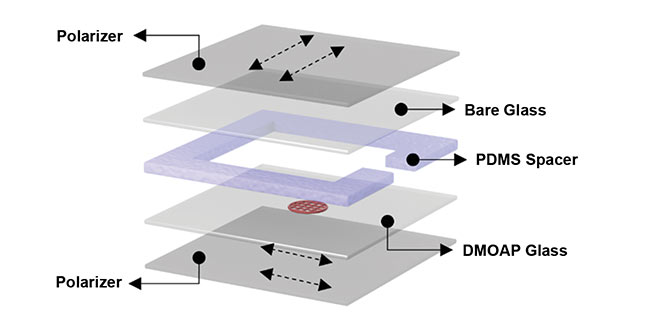
Figure 3. An exploded diagram of the layered makeup of the putative diagnostic device. The liquid crystal sensor on the DMOAP glass layer (red). PDMS: polydimethylsiloxane. Courtesy of professor Xiaoguang Wang/The Ohio State University.

Figure 4. Smartphone screenshots indicate a negative result (left) and a positive result (right). Courtesy of professor Xiaoguang Wang/The Ohio State University.
The next step for the COVID-19-related work is to move the test into a clinical evaluation phase — testing real patient samples in a biosafety Level 3 laboratory.
However, due to limited access to the labs throughout the pandemic, the group has been unable to perform these studies to date. Assuming it achieves satisfactory results at this next stage, and that it is able to validate the system in practical application, the group hopes to extend its collaboration to include the diagnostics industry for further studies and commercialization.
Meet the authors
Duncan Stacey has been the sales and marketing director for Linkam Scientific Instruments since 2014, working with leading companies in imaging, spectroscopy, and microscopy to develop the company’s products. He specializes in developing new markets and research solutions for temperature- and environmentally controlled experiments. Stacey is also a fellow of the Royal Microscopical Society and a member of its corporate advisory board; email: [email protected].
Xiaoguang (William) Wang, Ph.D., was recently appointed assistant professor in the William G. Lowrie Department of Chemical and Biomolecular Engineering at The Ohio State University College of Engineering, where his recent groundbreaking work in the field of SARS-CoV-2 took place. Wang was awarded his first-class honors and master’s degrees in chemical engineering in China before moving to the University of Wisconsin-Madison in 2016 to complete a doctorate with professor Nicholas Abbott. Wang’s research there focused on liquid crystal-templated assembly of colloids and molecules; email: [email protected].
Reference
1. Y. Xu et al. (2020). Ultrasensitive and selective detection of SARS-CoV-2 using thermotropic liquid crystals and image-based machine learning. Cell Rep Phys Sci, Vol. 1, Issue 12, www.doi.org/10.1016/j.xcrp.2020.100276.