New research includes assessment of capsid-based contrast agents as well as a method to detect the early stages of osteoarthritis.
Hank Hogan, Contributing Editor
MRI, which allows doctors and clinicians to peer inside the body without the use of ionizing radiation, is particularly useful for imaging soft tissues, which is why athletes often go into a scanner when injured.
Recent advances promise to make MRI an even better imaging tool. A look at research into nanosize contrast agents and naturally occurring arthritis markers reveals the possibilities, which could be a boon for patients undergoing an MRI scan.
For better MRI results, it helps to relax. That dictate applies not only to patients but also to the water protons inside them. During MRI, water protons are pushed out of alignment by properly tuned radio-frequency waves, and then they realign when exposed to a magnetic field. MRI detects the difference in water proton relaxation rates as this realignment happens. Thus, the greater the difference, the easier it is to distinguish between types of tissue or between diseased and healthy tissue.
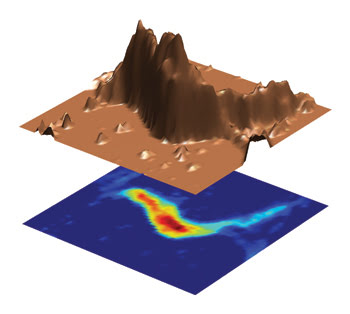
A technique researchers used for imaging glycosaminoglycan concentration in knee cartilage (pictured here) also might be used for diagnostic imaging of other conditions because glycosaminoglycans are found in intervertebral discs, heart valves and corneas. Courtesy of Alexej Jerschow, New York University.
Because relaxation rate differences in living tissue are usually small, contrast agents are used to boost the signal by increasing the relaxation rates.
Researchers from the University of California, Berkeley, from Università del Piemonte Orientale “A. Avogadro” in Alessandria, Italy, and from Lawrence Berkeley National Laboratory in California recently investigated contrast agents attached to capsids, which might one day lead to targeted MRI imaging with nanosize contrast agents.
“The ultimate goal is to synthesize contrast agents with high solubility, stability and relaxivities,” said Berkeley chemistry professor Kenneth N. Raymond.
Capsids, the protein coats of viruses, can be used as a scaffold for attaching contrast agents, in part because capsids larger than 10 nm are big enough to be retained in the blood under normal conditions but small enough to diffuse across endothelial barriers where pathology is present. In addition, the capsids have two surfaces — inside and outside — that allow contrast agents to be attached to one surface while the other one can be functionalized for targeting.
However, questions remain about the use of capsids and how they affect contrast agents. For instance, contrast agents need access to water because they work by changing the water proton relaxation rate. Thus, if diffusion of water into and out of a capsid isn’t fast enough, the MRI signal will suffer.
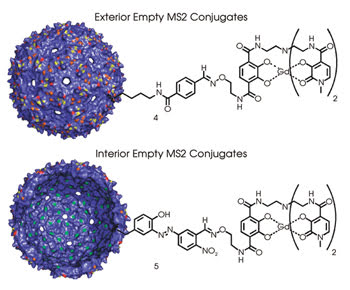
Researchers tested whether it was better to attach a contrast agent inside a hollowed-out MS2 capsid with a contrast agent attached to the outside (above) or to the inside of the viral shell (below). A total of 90 contrast agent molecules are attached in both cases. Reprinted with permission of the Journal of the American Chemical Society.
The researchers, therefore, conducted a detailed study of the effect of various parameters involved in making conjugates of contrast agents and capsid — information that will guide further development. They were able to do so because they had access to a field cycling relaxometer from Stelar srl of Mede, Italy. “This equipment is specifically designed to measure relaxivities at different field strengths, and there are only a few of these in the world,” Raymond said.
With this equipment, the researchers examined what occurred when they conjugated gadolinium chelates on the interior and exterior of bacteriophage MS2 capsids stripped of nucleic acids. They used gadolinium hydroxypyridonate-based contrast agents because they were seeking a relaxivity 20 to 100 times higher than what is now clinically available. These agents have demonstrated relaxivities that are double that of the desired threshold, performance achieved by having two to three bound water molecules and fast water-exchange rates.
The viral shell consisted of 180 copies of the coat protein assembled in an icosahedral arrangement, with a diameter of 27 nm. The researchers attached a linker to the gadolinium chelates and functionalized the capsids by modifying exterior amino groups or interior tyrosines. Approximately 90 chelates were attached per capsid.
Since they anticipate clinical applications, the researchers measured relaxivities at 298 and 310 K, just above room and body temperature, respectively. They found higher relaxivities for the internally attached contrast agents, with a maximum more than 33 percent higher than that of the externally attached contrast agents.
This better performance, they concluded, was related to the nature of the attachment. The internally attached agents were connected by a stiffer linkage than the externally attached ones, a difference that theory indicates would lead to higher relaxivities.
These results also showed that water diffused easily through the capsid. The researchers thought that this would happen, given the size of the pores in the viral shell. Thus, gadolinium hydroxypyridonate contrast agents attached to the inside of a viral capsid offered the best performance and did so while allowing the outside to be functionalized to target specific tissue or conditions. The work was published in the Feb. 27, 2008 issue of the Journal of the American Chemical Society.
While promising, these results are not as good as are needed, with relaxivities only 40 percent of what is desired. For that reason, the researchers are working on a new generation of capsid conjugates with better performance as contrast agents. Some of the changes involve new linkers while other modifications involve attachment strategies.
Raymond is hopeful that the work will yield the desired performance. “The hydroxypyridonate-based contrast agents have optimal water-exchange rates along with high relaxivities. This property will aid in attaining the high relaxivities that we are aiming for.”
He added that the next step will be to test the capsid agents in living tissue.
A new spin
For the tens of millions of osteoarthritis sufferers, a new MRI technique brings hope for an easier diagnosis and better monitoring of the degenerative joint disease. Developed by researchers from New York University and New York University School of Medicine along with researchers from Tel Aviv University in Israel, the technique maps the concentration of glycosaminoglycans. These molecules play a key role in cartilage health, and their loss is a characteristic of osteoarthritis.
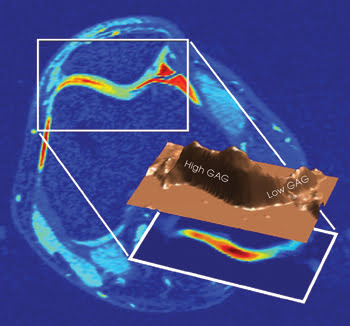
An MRI of a knee joint measures the glycosaminoglycan (GAG) concentration, which indicates the intactness of cartilage tissue. High concentrations are normal, whereas low concentrations are problematic. Such data someday could be used to assess the degree and progress of osteoarthritis. Courtesy of Alexej Jerschow, New York University.
In the past, MRI cartilage imaging has involved contrast agents that target glycosaminoglycans. However, this approach requires a long imaging time, hindering its use in a clinical setting. Another method that has been used depends on a determination of the concentration of the positively charged sodium that naturally occurs in cartilage. The sodium serves to map the negatively charged glycosaminoglycans that are also naturally present. Unfortunately, this technique has a low signal-to-noise ratio and requires special hardware — constraints that limit its clinical use.
However, the new MRI technique solves some of those problems. “The appeal of the method is that it does not require administration of contrast agents, and that it is applicable on standard equipment, which will make its use widespread,” said Alexej Jerschow, New York University associate professor of chemistry. He added that the technique could possibly be applied also to diagnostic imaging of other conditions, since glycosaminoglycans are found in intervertebral discs, heart valves and corneas.
In developing the method, the researchers applied a technique known as chemical-exchange-dependent saturation transfer. This procedure saturates exchangeable proton spins of a group of molecules, and that saturation is transferred upon chemical exchange to the bulk water pool. The result is an MRI contrast enhancement, with a billionfold boost in the signal reported under certain conditions. Thus, a relatively small signal from a few molecules can be detected easily. This is beneficial in the monitoring of glycogen concentration in the liver, for example.
In addition to applying chemical-exchange-dependent saturation transfer, the researchers took advantage of the structure of glycosaminoglycans. They are long unbranched carbohydrates with repeating disaccharide units, each of which has three hydroxyl (-OH) groups and one amide (-NH) group. Both groups are likely candidates for chemical-exchange-dependent saturation transfer, with -NH already proven useful in the mapping of brain proteins.
The researchers began by looking at the technique in the most basic way. “We first studied the effect in simple glycosaminoglycan solutions and degraded tissue samples,” Jerschow recalled.
In these studies, they irradiated solution and tissue samples using radio-frequency pulses to saturate the exchangeable proton spins. After irradiation, spectra taken with Siemens MRI hardware revealed that concentrations of 375 and 125 mM of -OH and -NH, respectively, caused a noticeable dip in the water signal intensity. The same features appeared in solution and in cartilage, although the cartilage spectra had additional features because of the more complex composition of cartilage.
To prove that the technique works for clinical diagnosis of osteoarthritis, the researchers studied living tissue. In fresh cartilage they examined the effect of introducing trypsin, a pancreatic enzyme that reduces the concentration of glycosaminoglycans. These tests showed that the hydroxyl signal served as the best chemical-exchange-dependent saturation transfer agent.
Finally, they imaged a human knee joint, finding regional variations in the concentration. The signal from the cartilage in the knee joint, for example, indicated a glycosaminoglycan relative concentration distribution of 33 percent, while the side and middle of the kneecap had concentrations of 22 and 18 percent. These variations were in line with those determined by other methods. The work was published in the Feb. 19, 2008 issue of PNAS.
Jerschow noted that applying the technique to live tissue was relatively straightforward. “It required some additional adjustments, such as fat saturation, and a limitation of the radio-frequency power supplied by the pulses, but otherwise was not difficult.”
He added that this approach, like other chemical-exchange-dependent saturation transfer methods, would benefit from enhanced magnetic field homogeneity. There are also ways to minimize the effects of inhomogeneities.
Jerschow said that the next step will be clinical studies. Images from patients with varying degrees of osteoarthritis will be taken, and the researchers hope to discover a way to correlate those images to the state of the disease.