A growing number of applications are driving development of optimized ultrafast laser sources with broader tuning, higher power, and streamlined ease of use.
DARRYL MCCOY AND MARCO ARRIGONI, COHERENT INC.
While multiphoton excitation is now firmly established as a front-line tool in microscopy, it is still an evolving field producing a steady stream of new applications and techniques, all supported by advancements in lasers and microscopes.
A key trend is the push for deeper imaging in vivo. It has recently been shown that a broadly tunable laser source enables researchers to use a “guide star” method, originally used in the field of astronomy, to perform the wavefront correction needed for high-resolution two-photon images deep in the mouse brain. Furthermore, researchers have discovered that three-photon excitation, using a new generation of amplified sources, enables deep imaging in dense tissue to help investigate the inflammatory response in mammalian lymph nodes. In addition, the maturation of multiphoton excitation has led to a broadening applications base that has created the need for compact, economical, fixed-wavelength ultrafast lasers that feature operational simplicity and high reliability.
Deep two-photon images
First-generation multiphoton micro- scopes relied on tunable titanium: sapphire (Ti:sapphire) lasers. These lasers proved ideal for exciting traditional dyes and calcium indicators optimized for single-photon excitation with blue-green light, as well as for green fluorescent protein (GFP) and its various derivatives. However, demand emerged for lasers widely tunable at longer wavelengths (950 to 1100 nm), particularly in neuroscience — for example, to use red-shifted fluorescent proteins such as the mFruit series developed by the late Roger Tsien.
Besides providing an extended palette of colors for multiple-stain imaging, long wavelengths also support deeper tissue imaging due to their reduced optical losses. Wide tuning enables rapid imaging of all the stains present in the sample. Practically, Ti:sapphire lasers and their accessories fall short of providing the 1 to 2 W of average power required for long-wavelength imaging through the 1-mm depth of the mouse cortex. The laser industry solved this challenge by developing tunable sources based on ytterbium (Yb) as the gain medium. The power and tuning range of these single-box sources continue to be increased, primarily to support deeper imaging in neuroscience based on two-photon excitation. An example of this type of laser is the Chameleon Discovery NX, which provides continuous tuning from 680 to 1320 nm with up to 3 W of output power.
Research from the David Kleinfeld laboratory at the University of California, San Diego provides an example of how the exploitation of these long wavelengths enables deep imaging in the mouse brain at unprecedented spatial resolution. One challenge for deeper imaging is that the effective resolution of two-photon imaging is negatively affected by optical aberrations, and the resolution declines with increasing depth.
“Two-photon excitation dictates the use of an optical system with high numerical aperture, so the microscope itself has some minor spherical and other wavefront aberrations,” said Rui Liu, a postdoctoral fellow in the Kleinfeld lab. “More importantly, mouse brain tissue is inhomogeneous with random variations in refractive index. As we probe deeper and deeper into this tissue with light focused through a high NA objective, the inhomogeneity causes wavefront distortions with the results that the excitation beam waist is far from being the ideal diffraction-limited circular spot.”
A solution to enable deep multiphoton imaging (inspired by wavefront correction when performing high-resolution astronomy from Earth-bound telescopes) was developed in the Kleinfeld lab. Specifically, a guide star created in the brain was used together with a Shack-Hartmann wavefront sensor (SHWS) to provide feedback signals that control the shape of an adaptive mirror (Figure 1). The SHWS consists of a 2D lens array that forms a dot pattern on a digital (electron multiplying CCD) camera. Nonuniform intensities of this pattern map wavefront distortions.
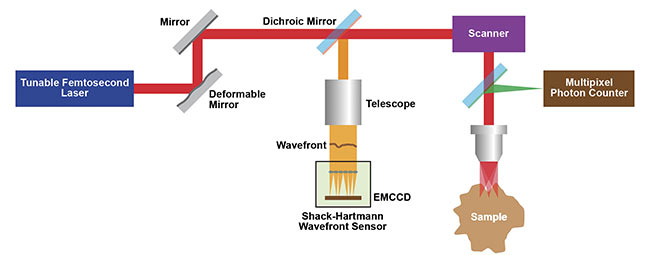
Figure 1. A schematic of the Kleinfeld lab’s wavefront correction 2P microscope setup that uses adaptive optics (AO) from a deformable mirror.
Courtesy of Kleinfeld lab.
“Broad laser tuning is a critical advantage here because it allows us to use light with a wavelength of 1.25 µm to excite the guide star based on the benign indicator Cy5.5-dextran introduced into the cerebral blood vessels,” Liu said. “We then use light at shorter wavelengths just above 1 µm to excite indicators (e.g., mRuby2, eYFP) of subcellular structure and function.” This avoids photobleaching of nonrenewable indicators during measurement of the tissue-induced aberrations. And, most importantly, all the excitation and emission wavelengths are within the deep penetration window (~650 to
1350 nm) sandwiched between the absorption peaks of hemoglobin and water.
The success of this guide star approach is illustrated in Figure 2, which compares images made of thalamocortical axons at a depth of 550 µm in a primary sensory cortex region of a live mouse, labeled by SF-Venus-iGluSnFR.A184S. The three images are recorded as follows: the first without invoking adaptive optics (no AO), the second with correction of just the microscope optics (system AO), and the third with correction of the entire imaging path including the tissue (full AO).
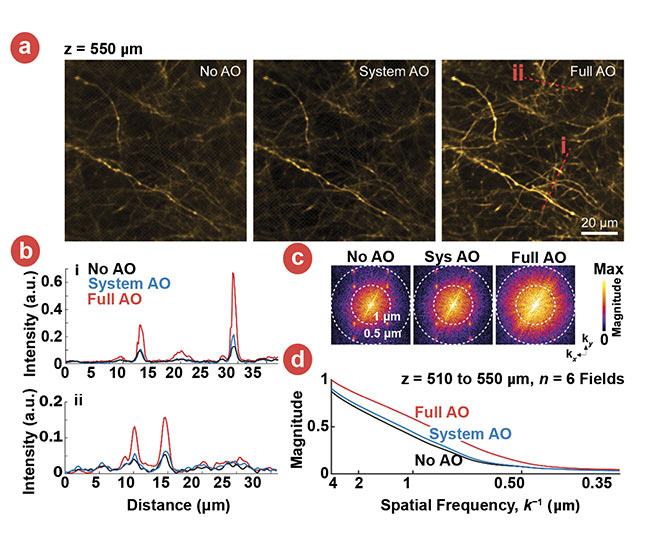
Figure 2. Images of thalamocortical axons in vS1 labeled by SF-Venus-iGluSnFR.A184S under the
conditions of no AO, system AO, and full AO (a). Signal profiles of the red lines in (a) are compared among no AO, system AO, and full AO (b). Spectral power as a function of spatial frequency, k, for the images in panels (a) (c). A k-space plot of the spatial frequency from the images of six fields at the depths of 510 to 550 µm below the pia at the conditions of no AO (black), system AO (blue), and full AO (red) (a). Courtesy of Kleinfeld lab.
Three-photon imaging for study
While the guide star method addresses the wavefront distortion and tissue absorption challenges of deep imaging, another method — three-photon (3P) excitation — is focused on increasing imaging depth by minimizing the light attenuation caused by scatter and absorption. This is a relatively new technique, pioneered by Chris Xu’s research group at Cornell and other universities, in which the laser wavelength is 3× the linear absorption wavelength rather than 2× as in two-photon excitation.
Since scattering in inhomogeneous tissue has a high order dependence on wavelength (roughly 1/λ4), a 50% longer wavelength can penetrate deeper in tissues. In addition, the higher nonlinearity of the three-photon effect means the fluorescence is excited in a small axial region close to the focal plane, resulting in better signal-to-noise ratio1.
Furthermore, 1300- and 1700-nm wavelengths match the 3P absorption spectrum of green- and red-shifted genetic expressions such as GFP, GCaMP, and tdTomato, and they fall within local minima of the brain tissue absorption spectrum. Three-photon excitation is thus useful in brain tissue to read neuronal signals in deeper cortex layers and even through the intact skull — that is, without the need of a cranial window. It can also be similarly valuable for intravital imaging in dense tissues with higher scatter levels, such as in bone, kidneys, lymph nodes, and spleen.
Three-photon excitation requires an advanced laser source and an improved microscope. Specifically, because the efficiency of 3P excitation relies on the cube of the focused laser intensity, the ideal laser source should have higher peak power (i.e., shorter pulse width) than a laser for 2P excitation, similar average power, and tunability around the 1300- and 1700-nm windows. These needs can now be met by combining a current-generation compact ytterbium ultrafast fiber amplifier with a tunable optical parametric amplifier (OPA), optimized for short (~50 fs) pulse width.
Until now, most published data from 3P excitation studies has been obtained using home-built microscopes looking at in vivo mouse brains. But the optics of commercial 2P microscopes does not have the requisite broad spectral range for 3P excitation. Recently, LaVision BioTec GmbH customized one of its TriM Scope microscopes to expand the spectral range of its input and scanning optics to deliver near diffraction-limited performance across the 700- to 1700-nm range. This microscope was used in professor Raluca Niesner’s laboratory in the German Rheumatism Research Center (DRFZ) in Berlin, together with the Monaco Opera-F laser and tunable OPA.
“One of the targets of my research group is improved understanding of the rogue inflammatory response that results in rheumatoid arthritis in particular, and in chronic inflammatory diseases in general,” Niesner said. “In addition, we are focused on developing imaging techniques to enable noninvasive interrogation with both high molecular specificity and high spatial resolution. These interests converge where we have been optimizing multiphoton imaging techniques to study the lymph nodes located near the joints of mammal (murine) limbs.”
Lymph nodes and the overlaying tissues are densely packed with cells, so deep 2P imaging has been quite challenging, according to technical director Volker Andresen of LaVision. “Imaging depth in dense tissues is often limited to a few hundred microns,” he said, “plus the effective resolution — the point spread function (PSF) — can decrease by an order of magnitude.”
The preliminary studies of limb lymph nodes by Asylkhan Rakhymzhan in Niesner’s lab indicate that 3P excitation can deliver a dramatic increase in deep imaging capabilities, specifically a >2× increase in maximum imaging depth for 3P versus 2P under identical conditions.
For example, one direct comparison combined multiphoton excitation of tdTomato fluorescence (expressed in blood vessels), backscattered second-harmonic generation (SHG) signals from collagen fibers, and third-harmonic generation (THG) of anisotropic structures such as lipid bilayers or collagen fibers. The maximum imaging depth was 300 µm based on 3P at a wavelength of 1630 nm, versus only 100 µm with 2P (at a wavelength of 1100 nm). Just as important, in these images, no noticeable decrease in spatial resolution occurs with increasing depth. Figure 3 shows typical images from a
z-stack recorded in this study.
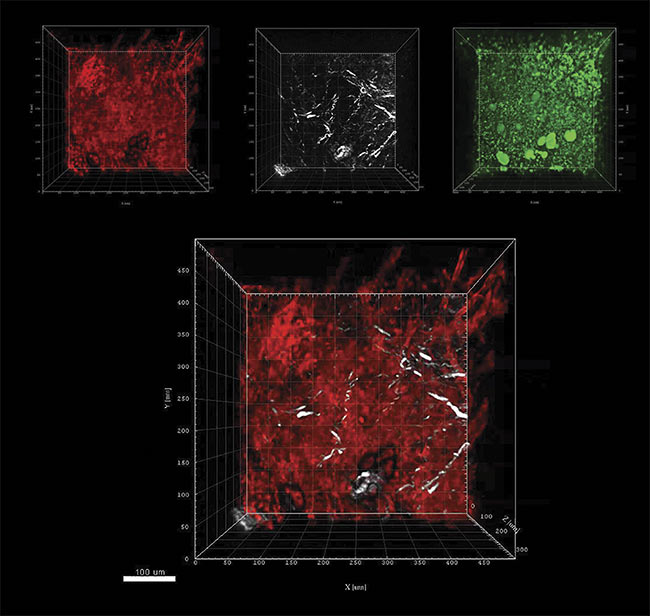
Figure 3. Murine lymph node z-stack images where z = 300 µm using a Coherent Monaco Opera-F combination tuned to a wavelength of 1630 nm,
and a LaVision TriM Scope microscope. Three-photon excitation of tdTomato detected at 595 (±20) nm (red); SHG signals from collagen fibers detected at
810 (±45) nm (white); and THG of lipid bilayers, lipids, or other anisotropic structures detected at 525 (±25) nm (green). Combined tdTomato and SHG signals (composite). Courtesy of Niesner lab.
Market needs streamlined lasers
While the latest tunable lasers and amplifier systems meet the needs of cutting-edge applications, at the other end of the multiphoton imaging market is a strong demand for simple, fixed-wavelength lasers at a lower cost. The need for these lasers is coming from three distinct segments of the market. First, the acceptance of multiphoton imaging as a tool rather than as a research field has created a demand across diverse applications for a turnkey laser with the operational simplicity of a light bulb. Second, applications such as optogenetics are driving the need for a second fixed wavelength — for example, from laboratories that already have a tunable Ti:sapphire laser. Third, end users and instrument makers are increasingly looking at multimodal tools in which multiphoton excitation/imaging is combined with another technology such as an atomic force microscope or even a flow cytometer. Ytterbium fiber is the gain medium of choice to meet this demand.
More than in any other scientific laser field, developments in multiphoton imaging applications are tightly interwoven with advancements in the lasers that support these applications. This close synergy among laser manufacturers, end users, and microscope suppliers is the key to success for cutting-edge applications and broad-use OEM instruments.
Meet the authors
Darryl McCoy is director of product marketing at Coherent Inc., managing the ultrafast tunable and fiber laser products for nonlinear microscopy and instrumentation markets.
Marco Arrigoni is director of strategic marketing at Coherent Inc. He covers the scientific research markets.
Reference
1. D. McCoy and M. Arrigoni (April 2018). Multiphoton microscopy sets the standard for live-cell imaging. BioPhotonics, Vol. 25, Issue 3, pp. 22-25.