Molecular imaging could help to advance clinical screenings for esophageal and other cancers.
Esophageal cancer is relatively rare: In 2013, only about 18,000 people in the US will be diagnosed with it, compared with the American Cancer Society’s projection of 1.7 million new cases of cancer generally this year. But esophageal cancer is an especially difficult one to manage. Symptoms – including difficulty swallowing and unexplained weight loss – typically occur late in the disease. By the time they appear, the cancer is often incurable. Because of its poor prognosis, it is one of the leading causes of death from cancer in the world.
Studies have demonstrated the many changes cells undergo as esophageal cancer develops, and such understandings can contribute to better management of the disease. “If we can identify early dysplasia, we can prevent the cancer from developing, or, if it’s already there, we can treat it much more effectively and less invasively,” said Elizabeth L. Bird-Lieberman, a University of Cambridge, UK, investigator who studies esophageal cancer.
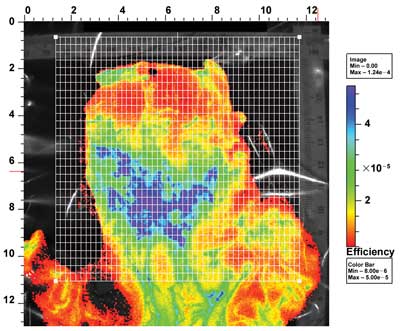
Researchers are developing a molecular imaging technique that can
help in the detection of esophageal cancer, based on changes in
lectin-binding patterns as the cancer progresses. Shown is an esophagus
that has been opened up and labeled with wheat germ agglutinin with a
fluorescent label. This demonstrated the different levels of binding
within the esophageal tissue (high binding is shown in blue, low in
red). Pathological examination then determined whether the binding
correlated with areas of dysplasia. Courtesy of Elizabeth
Bird-Lieberman, University of Cambridge.
This isn’t always so easy, though. Some dysplasia can be seen with the naked eye, but many others cannot. Today, patients with Barrett’s esophagus – often a precursor to esophageal cancer – regularly undergo endoscopies where samples are taken every 2 cm for biopsy. “This samples only a tiny area of the lining,” Bird-Lieberman said. “We’re often worried we might miss the dysplasia.”
We need new ways to identify such dysplasia, she said, and to find cancers earlier, even before symptoms appear.
Molecular imaging can give us a means to do so. Using this approach, researchers and clinicians can see intrinsic processes in cells and organs – including those associated with cancer – well before they are structurally evident. Thus, they can detect cancers much earlier than is possible with conventional imaging modalities.
“From a screening perspective,” said Sarah E. Bohndiek, a postdoc fellow in the Multimodal Molecular Imaging Lab at Stanford University in California, “we can look for abnormal changes in tissue – abnormal proliferation of cells, development of new blood vessels where they are not expected. Whereas, when looking at bulk anatomical changes, we can’t necessarily detect disease early in the time course.”
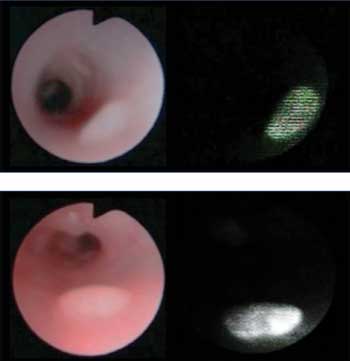
Optical molecular imaging can also aid in early detection of colon
cancer. Shown are endoscopic images of chemical-induced colon cancer in a
mouse model, including a white light image (left) and a fluorescence
image acquired using an activatable molecular probe. Courtesy of
Hisataka Kobayashi, NIH National Cancer Institute.
Identifying cancers at these very early stages – before they can be detected by radiological methods such as x-ray – can lead to greatly improved outcomes for patients diagnosed with cancer, increasing the odds of survival while reducing the cost and psychological toll of treatment.
Imaging epithelial cancers
Esophageal cancer offers a good model in which to study the progression of the disease. The esophagus is normally lined with cells called squamous cells. But when exposed to acid – as happens with gastroesophageal reflux disease (GERD) – these start to look more like the cells found in the stomach or small intestine. This stage is called Barrett’s esophagus. Each year, roughly 1 in 200 patients diagnosed with Barrett’s will go on to develop esophageal adenocarcinoma.
In a Nature Medicine study published last year, Bird-Lieberman and colleagues demonstrated that a certain type of sugar undergoes changes as Barrett’s esophagus progresses to adenocarcinoma, resulting in specific changes in lectin-binding patterns. The changes are not detectable with conventional endoscopy, but the researchers were able to see them by visualizing wheat germ agglutinin binding to the sugars. So the technique enables early detection of dysplasia in Barrett’s esophagus. Importantly, Bird-Lieberman said, they were able to image the changes using standard equipment “that everyone has in the clinic.”
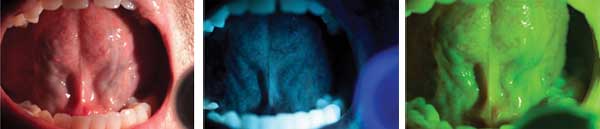
In some cases, molecular imaging of cancer relies on
endogenous contrast. The Identafi system, offered commercially by
DentalEZ Group and exclusively through Henry Schein, takes advantage of
the optical properties of tissue to detect biochemical and morphological
changes in the cells of the mouth, throat, tongue and tonsils that
could lead to oral cancer. The Identafi uses three wavelengths of light
as well as fluorescence and reflectance technologies to enhance oral
screening procedures. The dental professional first examines the oral
cavity with the “white light,” then switches to the “fluorescent violet
light.” This light helps identify areas of abnormal tissue; because the
violet light enhances normal tissue’s autofluorescence, suspicious
tissue will appear darker. If any abnormalities are found, the dental
professional will examine the oral cavity with the “Green-Amber
Reflectance Light,” which enables direct observation of the difference
between normal and abnormal tissue’s vasculature. Courtesy of DentalEZ
Group (DentalEZ acquired the technology from Trimira in 2011).

Topical application of the molecular probe is ideally suited to endoscopic imaging in epithelial cancers, including cancers in the esophagus and elsewhere in the gastrointestinal tract. There is a tremendous need for improved imaging in these cases. Some 80 percent of malignancies diagnosed every year are found on epithelial surfaces. And because the diagnosis is often made at a late, incurable stage, the cancers are associated with poor survival.
Hisataka Kobayashi, a researcher with the Molecular Imaging Program at the National Institute of Health’s National Cancer Institute (NCI) in Bethesda, Md., has been exploring the possibility of detecting colon cancer. In a 2012 paper published in Gut, he and colleagues at the NCI and the University of Tokyo described a rapid fluorescence detection method for use during colonoscopy. The method relies on a topically applied, enzymatically activatable probe that fluoresces when it is near γ-glutamyltranspeptidase, an enzyme associated with cancer.
As with Bird-Lieberman and others developing molecular imaging techniques for cancer screening applications, Kobayashi talked about how topical application of the molecular probe can lower the regulatory hurdles in translating the methods to the clinic. Generally, he said, making the early detection techniques quick, inexpensive and potentially nontoxic will get them to the clinic faster. Once there, the techniques will help find cancers sooner than is typically possible with conventional imaging modalities.
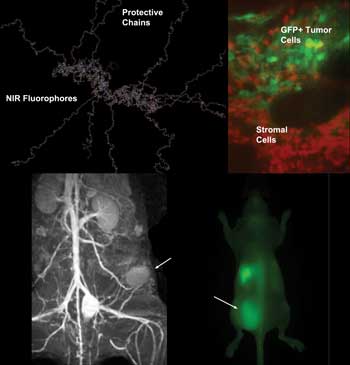
Using molecular imaging, Alexei Bogdanov Jr. and colleagues
observed that tumor-supporting cells – as opposed to the cancers
themselves – were the most actively involved in uptake and degradation
of a near-infrared fluorescence probe, PGC. Here, fibroblasts are
near-infrared fluorescent because of PGC cleavage, and they outnumber
tumor cells, which are expressing GFP and produce red fluorescence. A
1999 Nature Biotechnology study by Ralph Weissleder, Ching-Hsuan Tung,
Umar Mahmood and Bogdanov was among the first to demonstrate the
potential of in vivo molecular imaging of cancer in animal models. Shown
is a previously unpublished image captured with this technique. Images
courtesy of Alexei Bogdanov, University of Massachusetts Medical School.
New cancer-screening paradigms
Ultimately, these techniques could be one part of a broader clinical strategy. Bohndiek, the Stanford investigator, said that she and her colleagues – and other research groups – are seeking to establish a paradigm in which molecular diagnostics begins with blood panel screening. If a patient’s blood biomarkers indicate the possible presence of cancer, then he or she is brought in for an imaging test to learn more. This makes sense from both a cost and time perspective, and of course from the perspective of reducing patient stress.
Down the road, it may be possible to find cancers even sooner than with these molecular diagnostics approaches. “What we really want to do is early-early detection,” Bohndiek said, “not just speculative testing of a population, but more of a predictive screen.” For example, deeper understandings of the genetic changes underlying cancer could enable genetic screening of a population.
Imaging in animal models
Preclinical imaging of cancer in animal models has not only spurred the development of new imaging strategies for cancer detection, it has also contributed to our understanding of how cancer arises and spreads. This new knowledge has advanced the rate of drug discovery and development, and allowed a new focus on individualized treatment.
Use of optical fluorescence techniques for such imaging dates back to the late 1990s. One of the first demonstrations of the potential of in vivo fluorescence imaging was shown in a study led by Ralph Weissleder at Massachusetts General Hospital in Boston and published in Nature Biotechnology. Alexei Bogdanov, who co-authored the study, described tumors in mouse models “lighting up” after exciting fluorescence in specialized autoquenched near-infrared fluorescence probes. The probes were based on a contrast agent, protected graft co-polymer (PGC), that Bogdanov had developed several years before to visualize the blood supply of tumors using MRI in animals.
Using these probes and others like them, researchers have gleaned important information about the development of cancer. Bogdanov and colleagues observed early on, for example, that in many cases, tumor-supporting cells – and not the cancer itself – were most actively involved in PGC uptake and degradation. Thus, optical fluorescence could prove essential for detecting early signatures of metastasis by allowing visualization of noncancerous stromal cells in potential sites of metastasis.
Since they were first demonstrated, PGC-based imaging probes have been used in a number of research studies aimed at improving cancer detection and treatment. Just as exciting is the potential for these probes to translate into clinical use.
Using “activatable” probes similar to PGCs, according to Bogdanov, who is now a professor of Radiology and Cell Biology at the University of Massachusetts Medical School, surgeons may be able to locate tumors in the body quickly and with high precision using intraoperative fluorescence imaging. “PGC gives a very low signal in its intact state, but its products are brightly fluorescent, which greatly aids in detecting tumors.” He added that the PGC carrier has already been tested in a clinical trial, so translation to the clinic could very well be within reach.