Fluorescent nanomaterials allow for imaging membrane potential at single-cell resolution while measuring data from thousands of cells at the same time.
BETHANY ALMEIDA, IGOR L. MEDINTZ, AND JAMES B. DELEHANTY U.S. NAVAL RESEARCH LABORATORY CENTER FOR BIOMOLECULAR SCIENCE AND ENGINEERING
Despite increasing efforts to solve the puzzle that is the workings of the human brain, scientists’ current understanding of the brain’s functional interconnections remains limited. As the most complex organ in the human body, the brain is an immense network of neuronal cells capable of sending signals to receptors in the rest of the body. These signals begin as chemical changes, such as rapid variations in ion concentration through sodium and potassium (Na+/K+) ion channels in the neuronal cell membranes, which in turn result in electrical potential changes across the plasma membrane (Figure 1a)1,2.
Cadmium sulfide quantum dots. Courtesy of iStock.com/OlgaReukova.
Notwithstanding the enormous number of cells (~1010) and cell-to-cell connections (~1014) in the brain, multiple projects are currently underway to elucidate functions of the brain at both the nano and macro scales. Prominent efforts include the EU’s Human Brain Project and the U.S.’s BRAIN (Brain Research through Advancing Innovative Neurotechnologies) Initiative.
To visualize and record specific operations of the brain, scientists must be able to image cells with single-cell resolution and on a millisecond timescale while concurrently retaining the ability to measure thousands of cells. These twin goals are more reachable than ever before with the nanotechnology available in quantum dots (QDs), whereas conventional methods pose significant challenges when used to attempt to match this success.
The most widely used conventional method in the research laboratory setting is the patch clamp technique, in which two intracellular electrodes maintain membrane voltage at a set value1,2. However, this technique is invasive and cannot probe many cells at once. While other options such as electroencephalography and magnetoencephalography are noninvasive and exhibit millisecond time resolution, these methods are still limited by a lack of single-cell resolution1,3. Finally, voltage-sensitive dyes (e.g., FluoVolt, oxonol, and ANEPPS) are limited by their reliance on genetic engineering of cells for their expression, small changes in fluorescence during changes in membrane potential, and photobleaching (a loss in fluorescence over time due to light exposure)1-3.
Emerging interest in QDs
Although not the only type of nanomaterial currently being investigated for neuronal imaging, recent reports have demonstrated that luminescent, nanocrystalline, semiconductor QDs are potentially able to meet the stringent resolution requirements of neuronal imaging as a result of their unique photophysical and electronic properties (Figure 1b)1-3. QDs, often similar in size to the thickness of the plasma membrane (~5 nm), demonstrate facile bioconjugation (the ability to form a stable link) with membrane-targeted peptides or other moieties4,5. Furthermore, QDs have high photoluminescence, or emission of light upon excitation with photons, which can be modulated via an applied electric field, and they are not as susceptible to photobleaching as voltage-sensitive dyes1,2. The photoluminescence of QDs is directly associated with the size of the QD, which allows for multiplexed/multicolor labeling and can potentially enable the simultaneous imaging of different neuronal cell types and processes. Finally, QDs demonstrate the potential for deep brain imaging via their large two-photon absorption cross sections and excited-state lifetimes in the nanosecond regime (~20 ns)4,6.
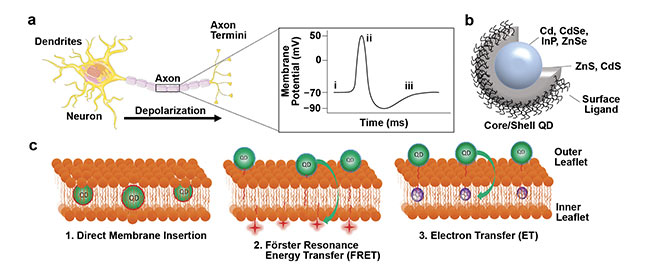
Figure 1. Mechanisms of QD interactions with the plasma membrane for imaging membrane potential. Schematic of a depolarizing neuron (a, left) resulting
in changes in membrane potential over time (a, right). Schematic of a ligand-coated core/shell QD (b). QDs can be interfaced with the plasma membrane by
1) direct membrane insertion, 2) FRET, and 3) electron transfer (c). Adapted with permission from Reference 2.
Initial studies by the Shimon Weiss group described the photoluminescence response of QDs to an applied electric field. In these studies, the researchers compared type I, type II, and quasi-type- II QDs. For QDs, “type” refers to the band alignment and electron/hole location within the core/shell structure, which results in distinct emission behavior for various QDs. From these studies, asymmetric type II and quasi-type-II QDs exhibited the greatest shift in quantum-confined Stark effect properties, which is typically characterized by spectral broadening, photoluminescence quenching, and a redshift of the emission maxima7. Furthermore, the sensitivity of neuronal spiking to an applied electric field was found to be influenced by the material composition of the QDs8.
To determine the sensitivity of QDs loaded within a neuronal plasma membrane, Clare E. Rowland and colleagues developed a platform wherein QDs were cast within a poly(methyl methacrylate) layer between two SiO2 dielectric layers9. When exposed to an electric field emulating that of a firing neuron, both type I and quasi-type-II QDs demonstrated photoluminescence quenching of ~2% and ~5%, respectively, and this quenching tracked temporally with the voltage trace.
QD-mediated neuronal imaging
There are three possible approaches to imaging neuronal cellular membrane potential with QDs (Figure 1c)1,2:
1. QDs may be inserted directly within the plasma membrane bilayer for direct measurement of the electrical field. They may be appended to the cellular membrane surface via membrane-targeting peptides with which they engage.
2. QDs can engage in Förster resonance energy transfer (FRET) with a proximal dye acceptor/quencher.
3. QDs can engage in electron transfer with a suitable electron acceptor.
Building on the theoretical, proof-of-concept studies detailed above, a handful of studies have been published that have tested the sensitivity of these QDs for neuronal membrane potential imaging both in vitro and in vivo. These studies are discussed below, organized by the specific QD-plasma membrane interaction strategy that is employed.
Direct membrane insertion
The most popular strategy among researchers is direct membrane insertion of QDs. The Weiss group published the first study using the membrane insertion strategy, building upon its previous theoretical analysis to develop quasi-type-II CdSe-seeded CdS QD nanorods decorated with peptides consisting of both hydrophobic (repelled by water) rigid α-helical domains and hydrophilic (mixed with water) domains designed to impart both hydrophilic and lipophilic (dissolving in fat) regions to the QDs (Figure 2a)10. As a result of their unique structure, the QDs were inserted into the plasma membrane in various orientations. Vertical/membrane-spanning insertion was observed for ~58% of events, proper horizontal insertion was observed for ~16%, and partial or tilted insertion was observed for ~41% (Figure 2b).
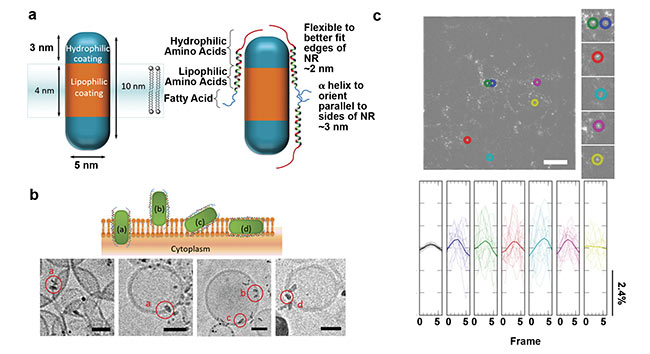
Figure 2. Imaging membrane potential using QD nanorods (NRs) directly inserted into the membrane. Quasi-type-II CdSe-seeded CdS QDs decorated with
a peptide (a). Schematic of QDs inserted into the plasma membrane in various orientations (b, top) and cryoEM micrographs of the QDs inserted into small
unilamellar vesicles (b, bottom). High-pass-filtered image of human embryonic kidney cells labeled with QDs (colored circles, c, top) and representative temporal, bandpass-filtered ΔF/F0 traces for each region of interest (c, bottom). Adapted with permission from Reference 10.
The reported voltage sensitivity of ensemble QDs inserted into human embryonic kidney cell membranes was a ~0.6% change in fluorescence intensity from the baseline (ΔF/F0) (Figure 2c). However, for those QDs exhibiting proper orientation, ΔF/F0 increased to ~5%. Further, controlling QD concentration resulted in individual or small clusters of QDs, potentially allowing for single-particle voltage imaging at discrete membrane locations.
This same group improved upon this design and developed voltage-sensing, seeded type II ZnSe/CdS QDs functionalized with both α-helical peptides and zwitterionic lipoic acids11. After one hour, these QDs demonstrated membrane localization, while similar positively or negatively charged QDs were endocytosed. Despite ensemble results for QDs inserted into human embryonic kidney cells resulting in only ~1% ΔF/F0, these results were statistically significant. The voltage-sensitive QDs demonstrated reversible voltage response, and photoluminescence was increased as opposed to quenched.
Finally, the Ulrich F. Keyser group compared the effects of traditional CdSe/CdS QDs with the more biocompatible (non-Cd-containing) InP/ZnS QDs12. Here, QDs were injected into Xenopus laevis (African clawed frog) retinal ganglion cells and not necessarily localized to the plasma membrane. Despite this, both QDs demonstrated an increase in photoluminescence during depolarization (ΔF/F0 of ~30% to ~60% for CdSe/CdS QDs and ~11% for InP/ZnS QDs).
FRET
Guangcun Chen and colleagues designed the only reported QD system for imaging neuronal membrane potential based on FRET5. In this study, the CdSe/ZnS core/shell QDs (as the FRET donor) were localized to the outer leaflet of the membrane bilayer of human embryonic kidney cells via the glutathione cap on the QD surface. The lipophilic anion dipicrylamine (DPA, a nonfluorescent membrane dye commonly used as a FRET acceptor/quencher) was also incorporated into the plasma membrane, allowing the QD and DPA to engage in FRET as a donor-acceptor pair, respectively. During depolarization, DPA translocates to the inner leaflet of the membrane bilayer, decreasing the FRET efficiency between QD and DPA, resulting in an increase in QD photoluminescence of ~4% to ~6%. A control experiment without the DPA acceptor showed no change in photoluminescence during depolarization.
Electron energy transfer
In a study by the James B. Delehanty group, quasi-type-II CdSe/CdS/ZnS core/multishell QDs were decorated with peptide-fullerene (C60) moieties, in which the QD and C60 engaged in electron transfer as donor and acceptor, respectively (Figure 3a)4. The quasi-type-II QD affords a more delocalized electron structure in the excited-state QD such that it can engage in efficient electron transfer with the C60. The peptide-C60 inserted into the membrane bilayer while the hydrophilic QD decorated the outside of the cell. In cervical cancer cells, three peptides of varying lengths were tested to determine how distance influenced electron transfer to modulate QD photoluminescence in response to changes in membrane potential, and it was found that the shortest peptide resulted in the greatest ΔF/F0 (~20%) compared to longer peptides. This short peptide resulted in a ΔF/F0 per mV equivalent to that of the fast-response voltage-sensitive dye FluoVolt (Figure 3b).
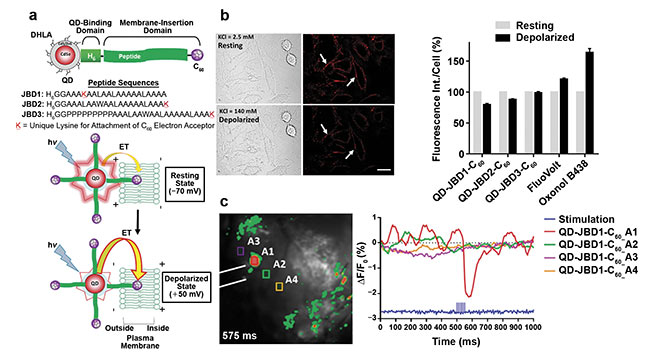
Figure 3. Imaging membrane potential using QDs appended to the plasma membrane. Schematic of CdSe/CdS/ZnS core/multishell QDs conjugated to
peptide-C60 (a, top). Schematic representation of QD-mediated electron transfer during changes in membrane potential (a, bottom). Differential interference contrast and confocal fluorescence images of A549 (cervical cancer) cells labeled with QD-peptide (b, left) and fluorescence intensity changes compared to commercial voltage-sensitive dyes (b, right). Representative frame of mouse cortexes injected with QD-peptide-C60 conjugates during electrical stimulation with four highlighted regions of interest (ROIs) (c, left). Time-resolved ΔF/F0 changes of the four ROIs depicted as an average of 50 trials (c, right). Adapted with permission from Reference 4.
Furthermore, the researchers demonstrated the sensitivity of this system by showing that QD-peptide conjugates (without the C60) resulted in less than 3% quenching of photoluminescence. In vivo, solutions of QD-C60 injected into mouse cortex resulted in ~2% ΔF/F0 upon stimulation with a tungsten electrode (Figure 3c). This was the first study to demonstrate a QD-bioconjugate system for imaging membrane potential during neuronal firing in vivo, with ~20- to ~40-fold larger ΔF/F0 compared to commonly used voltage-sensitive dyes.
Future perspectives
Despite the impressive advancements made in this field within the last five years, challenges remain that must be overcome to truly harness the potential of QDs for neuronal imaging. These include:
1. Improving the targeting of the QDs to particular subsets of neurons, ensuring that the QDs remain membrane resident, with minimal endocytosis.
2. Limiting the toxicity of some QD formulations.
3. Increasing the scale and multifunctionality of QD systems.
Furthermore, detection of spectral shifts associated with the quantum-
confined Stark effect can be slow. To address this point, a recent theoretical study from the Dan Oron group suggests the possibility of a detection mechanism to achieve shot-noise-limited sensitivity to emission wavelength shifts on a millisecond timescale13. Further, development of multifunctional QDs (and other nanoparticles) for noninvasive interfacing with neuronal cells, such as those described above, will facilitate the bridging of the man-machine interface — the seamless intercommunication between neurons in the human brain and electrical devices.
Consider, for example, the exciting goal of the current Next-Generation Non-Surgical Neurotechnologies (N3) program at DARPA, which is to noninvasively (wirelessly) record from ∼106 neurons while simultaneously being able to stimulate ∼105 neurons in a defined tissue volume (∼16 mm3). Advancements such as these will result in the implementation of technologies that are capable of wirelessly connecting the human brain with computers, medical devices, and prosthetics. And people can expect QDs and other nanoparticle materials to play a critical role in this endeavor.
Meet the authors
Bethany Almeida, Ph.D., received her doctorate in biomedical engineering from Brown University. She is currently an American Society for Engineering Education Postdoctoral Research Fellow in James B. Delehanty’s research group at the U.S. Naval Research Laboratory’s Center for Biomolecular Science and Engineering.
Igor L. Medintz, Ph.D., received his doctorate in molecular, cellular, and developmental biology from the City University of New York and completed postdoctoral research with Richard A. Mathies at the University of California, Berkeley. He currently serves as the Navy’s senior scientist for biosensors and biomaterials at the U.S. Naval Research Laboratory’s Center for Biomolecular Science and Engineering.
James B. Delehanty, Ph.D., received his doctorate in molecular and cellular biology from Tulane University. He is a Research Biologist IV at the U.S. Naval Research Laboratory’s Center for Biomolecular Science and Engineering. Delehanty is also an adjunct professor in the Fischell Department of Bioengineering at the University of Maryland.
References
1. A.L. Efros et al. (2018). Evaluating the potential of using quantum dots for monitoring electrical signals in neurons. Nat Nanotechnol, Vol. 13, pp. 278-288.
2. O.K. Nag et al. (2020). Nanoparticle-mediated visualization and control of cellular membrane potential: strategies, progress, and remaining issues. ACS Nano, Vol. 14, Issue 3, pp. 2659-2677.
3. K. Park and S. Weiss (2017). Design rules for membrane-embedded voltage-sensing nanoparticles. Biophys J, Vol. 112, pp. 703-713.
4. O.K. Nag et al. (2017). Quantum dot-peptide-fullerene bioconjugates for visualization of in vitro and in vivo cellular membrane potential. ACS Nano, Vol. 11, Issue 6, pp. 5598-5613.
5. G. Chen et al. (2019). Glutathione-capped quantum dots for plasma membrane labeling and membrane potential imaging. Nano Res, Vol. 12, Issue 6, pp. 1321-1326.
6. B.K. Andrásfalvy et al. (2014). Quantum dot-based multiphoton fluorescent pipettes for targeted neuronal electrophysiology. Nat Methods, Vol. 11, Issue 12, pp. 1237-1241.
7. K. Park et al. (2012). Single molecule quantum-confined Stark effect measurements of semiconductor nanoparticles at room temperature. ACS Nano, Vol. 6, Issue 11, pp. 10013-10023.
8. J.D. Marshall and M.J. Schnitzer (2013). Optical strategies for sensing neuronal voltage using quantum dots and other semiconductor nanocrystals. ACS Nano, Vol. 7, Issue 5, pp. 4601-4609.
9. C.E. Rowland et al. (2015). Electric field modulation of semiconductor quantum dot photoluminescence: insights into the design of robust voltage-sensitive cellular imaging probes. Nano Lett, Vol. 15, Issue 10, pp. 6848-6854.
10. K. Park et al. (2018). Membrane insertion of — and membrane potential sensing by — semiconductor voltage nanosensors: feasibility demonstration. Sci Adv, Vol. 4, Article e1601453.
11. J. Park et al. (2019). Improved surface functionalization and characterization of membrane-targeted semiconductor voltage nanosensors. J Phys Chem Lett, Vol. 10, Issue 14, pp. 3906-3913.
12. M. Caglar et al. (2019). All-optical detection of neuronal membrane depolarization in live cells using colloidal quantum dots. Nano Lett, Vol. 19, Issue 12, pp. 8539-8549.
13. O. Bar-Elli et al. (2018). Rapid voltage sensing with single nanorods via the quantum confined Stark effect. ACS Photonics, Vol. 5, Issue 7, pp. 2860-2867.