
Scientists harness photoacoustic effect for imaging
Technique could be useful for trauma evaluation
Kevin Robinson
Researchers at Texas A&M University in College Station have developed
a method for using the photo-acoustic effect to create images. The technique permits
functional imaging of oxy- and deoxyhemoglobin, which could be useful for trauma
evaluation, for example, where optical absorption reveals information related to
hemorrhage and edema. It provides axial resolution of about 15 μm, lateral
resolution of 45 μm and an imaging depth of 3 mm.
“This technique measures optical contrast
based on physiological parameters, such as the total hemoglobin concentration and
the oxygen saturation of hemoglobin,” explained Lihong V. Wang from the optical
imaging laboratory at the university.
The photoacoustic effect is an ultrasonic
wave created when tissue is irradiated by a short-pulse laser. Some of the radiation
is absorbed and converted partially into heat, which then creates a rise in pressure
through thermoelastic expansion. The pressure rise moves through the tissue as a
sound wave that can be detected by ultrasonic transducers.
“Since ultrasonic waves are much
less scattered than optical waves, photoacoustic imaging combines the contrast
of optical absorption with the spatial resolution of ultrasound,” Wang said.
The technique is a cross between absorption spectroscopy and ultrasound.
The researchers picked the absorption
band of hemoglobin and exposed tissue to laser light that fell into that band. Then
they used the ultrasonic wave to create an image. Ultrasound waves scatter 100 to
1000 times less than light waves, Wang said. “As a result, ultrasound can
provide much better resolution than light can for structures located deeper than
1 mm below a tissue’s surface.”
To create a functional photoacoustic
microscope, the investigators used a tunable dye laser that generated 6-ns pulses
and that was pumped by an Nd:YAG laser. The laser beam traveled to the scanning
head via an optical fiber, and a photodiode calibrated the laser energy. The beam
passed from the fiber through a conical lens that made a ring-shaped illumination
pattern. The ring was focused into the tissue where the focal area over-lapped
the ultrasonic focal area, although the optical focus was wider. The ring-shaped
pattern was intended to reduce the photoacoustic effect generated in the field of
view of the ultrasonic transducer. The system was set up to detect the laser-generated
photoacoustic signal in reflection mode.
Achieving maximum depth
The focal diameter of the ultrasonic transducer
determined the system’s lateral resolution. “If the laser pulse is sufficiently
short, a high-numerical-aperture acoustic lens and a high-center-frequency ultrasonic
transducer provide high lateral resolution. A wideband ultrasonic transducer provides
high axial resolution,” Wang said.
According to Wang’s paper, published
in the July issue of Nature Biotechnology, when the center frequency of the
transducer exceeds 10 MHz, the penetration depth of the ultrasonic wave —
not the penetration of the excitation light — determines the maximum imaging
depth. The researchers used a Panametrics 6-mm ultrasonic detector with a 50-MHz
central frequency and a 70 percent nominal bandwidth. Coupling the device to a homemade
spherically focusing lens with a numerical aperature of 0.44, a focal zone of 0.3
mm and a focal length of 6.7 mm, the group created images with an axial resolution
of 15 μm and a lateral resolution of 45 μm at a depth of up to 3 mm in
living tissue.
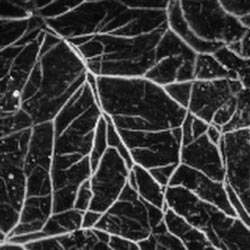
This in vivo image shows subcutaneous microvasculature from a 20-g SENCAR mouse at a
584-nm optical wavelength. SENCAR mice are commonly used for experiments with carcinogens.
Hemoglobin and melanin are responsible
for most optical absorption that creates the photoacoustic effect. As a result,
choosing the correct excitation wavelength permits blood detection, which the researchers
say enables high-contrast, specific images of the microvasculature.
At 584 nm, the laser emitted the wavelength
where oxygenated and deoxygenated hemoglobin had the same molecular extinction coefficient.
As a result, the image contrast became dependent on the total concentration of hemoglobin
but did not reflect changes in oxygenation levels. This could be useful in imaging
the blood vessels that characterize rapidly growing tumors and could aid in evaluating
the effectiveness of therapies that target angiogenesis.
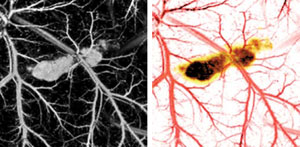
In vivo images of a five-day postinoculation melanoma tumor in a
20-g immunocompromised nude mouse were taken at 584-nm (left) and 764-nm optical
wavelengths. A pseudocolored composite of the images from the two wavelengths is
shown on the right.
The group demonstrated the same technique
with dual-wavelength imaging. They used 584-nm light to image blood vessel proliferation
in a tumor and then re-imaged the same section with 764-nm light that missed the
absorption peaks of hemoglobin and melanin and penetrated deeply into the tumor,
providing information about the thickness of a particular tumor. Combining the two
images created a high-contrast image capable of resolving individual microvessels
roughly 50 μm in diameter. In addition, the dual-wavelength technique can measure
oxygen saturation in individual blood vessels.
The researchers first tested the oxygen
saturation measurements ex vivo using bovine blood and compared their results with
a standard optical oxygen saturation measurement. When the results proved to be
similar, they used the system to measure normal oxygen saturation in live human
tissue, detecting a saturation level of 97 ±2 percent for arterial blood, and 77
±4 percent for venous blood. They also demonstrated that the system can respond
accurately to a shift from normal oxygenation to hypoxic conditions. The system
not only created an accurate measurement of overall oxygen saturation, but also
can map the saturation levels of individual blood vessels. The group thinks that
this that may be useful in conducting functional brain imaging.
At present, however, the system is
somewhat slow. While a single, one-dimensional depth image takes only about 2 μs,
a one-dimensional scan in both the depth and the transverse directions takes about
10 s. It takes a full 18 min to get a two-dimensional image of a 64-square-mm area,
and more than 2 hours for the oxygen saturation measurements. Wang and his colleagues
acknowledged this and posit that, by increasing the laser repetition rate and using
an array of ultrasonic transducers, they can make the system faster.
They plan to begin studying the new
method in clinical applications such as melanoma imaging, Wang said.
Wang’s laboratory has moved to
Washington University in St. Louis.
Published: September 2006