
Spectroscopic Technique Targets Drug Porosity
Terahertz time-domain method establishes tablet effectiveness with contact-free measurement.
By Thomas Juliano
In the pharmaceutical field, porosity is an important trait in helping to measure the effectiveness of tablet quality. Thanks to modern spectroscopic techniques, manufacturers are getting the characteristics they’re looking for. Porosity, in addition to other properties such as surface area, particle size, and solubility, is a primary contributing factor in the dissolution rate of tablets. Through this attribute, producers and researchers are able to obtain an initial indication of liquid absorption and, therefore, dissolution properties of pharmaceutical tablets.
Generally speaking, the higher the porosity, the faster the absorption of liquid into a matrix or composite material1. The absorption of liquid into a tablet matrix enables the contents to begin to swell, which eventually leads to the tablet physically breaking down. Because the active pharmaceutical ingredients (APIs) are located inside the tablet, the tablet must be broken down to deliver the APIs.
Typically, the porosity is reported mathematically as the fraction of the volume of voids, or space, over the total volume of the tablet2. It is crucial to understand this characteristic of tablets because it can play a large role in the disintegration process within the tablet3. This feature is affected by both the compression speed and compaction forces used when forming tablets1. Therefore, the ability to rapidly monitor changes in these parameters would be a powerful tool.
Porosity is a volumetric property of a material that governs the mass transport within materials. In this example, however, the transportation of water through the tablet matrix can be applied to other materials that are dependent on this property, such as catalysts.
The traditional method for determining the porosity of a tablet, porosimetry, measures it directly by gas or liquid intrusion4. This technique is slow, however, and can be destructive to the medicine being produced. One typical porosimetry method uses mercury as the liquid introduced into the pores of the sample. This method depends on the intrusion of mercury into the pore system of the samples under a controlled pressure environment. Once the mercury has been forced into the pores of the sample, the sample is not recoverable; therefore, any product tested using this method is marked as a loss.
Recent works have shown that terahertz time-domain spectroscopy (THz-TDS) could be a viable method for nondestructively measuring the porosity of pharmaceutical tablets5,6. This was first shown for flat-faced tablets and later expanded to biconvex tablets5,7. THz-TDS uses the setup shown in Figure 1.
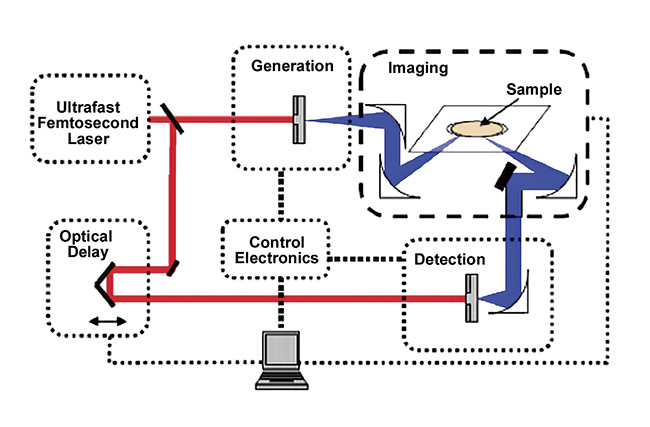
Figure 1. A schematic of a terahertz time-domain spectrometer setup. Courtesy of TeraView Ltd.
Early on, porosity measurements using THz-TDS were taken on flat-faced tablets. The tablets used by Prince Bawuah and colleagues were compressed and evaluated in the lab. The researchers were able to control conditions such as pressure, time, weight, and thickness5. After they established that the THz technique showed promise, it was applied to a wider range of tablets, including ones that were biconvex. These tablets contained both excipients and APIs and were measured using THz-TDS, similarly to the method used for flat-faced tablets7. Once the method was shown to be effective for evaluating sample sets produced in research lab conditions, THz-TDS was then applied to sample batches produced using a production-scale method1.
THz-TDS has several advantages over older, more traditional methods: It is noncontact and nondestructive, and it can measure a large amount of product in a short amount of time. The typical acquisition time for THz waveforms is 15 Hz (15 waveforms per second), meaning a sample set of 60 waveforms can be acquired in
~4 s. Averaging the measurements allows for a reduction in the spectral noise. While THz-TDS was shown to be effective on biconvex tablets, another advantage was shown by the researchers: No sample preparation is needed. The tablet can be placed directly into the THz-TDS transmission chamber and the measurements can be taken quickly.
Terahertz waves are located on the electromagnetic spectrum between microwave and infrared, from about 60 GHz to 10 THz. This range has unique properties that make it very useful in sample analysis. Terahertz waves are nonionizing and can penetrate deep into dielectric materials, but the penetration depth is material specific. The depth is also dependent on wavelength, and the higher frequency of the waves of terahertz pulses allows them to penetrate deeply into most pharmaceutical materials. This is critical to the effectiveness of terahertz porosity measurements. Samples can be placed into the terahertz transmission setup, measured, and then recovered without sustaining damage1.
Equipment setup
To acquire the terahertz data used to determine the porosity of tablets, the proper equipment is needed for measurement of the product. Using a transmission spectroscopy setup (Figure 1) with a benchtop laboratory spectrometer (Figures 2 and 3), the sample (tablet) is placed between a terahertz emitter and a receiver. The terahertz beam passes through the entire tablet. As long as the tablet thickness is known (measured beforehand), the terahertz data can be analyzed to provide the effective refractive index of the sample.

Figure 2. A terahertz transmission spectroscopy chamber. Courtesy of TeraView Ltd.

Figure 3. A TeraView TeraPulse Lx benchtop laboratory terahertz spectrometer. Courtesy of TeraView Ltd.
The porosity is related to the effective refractive index of the samples and can be determined from these measurements. The greater the number or size of the pores within a material, the lower its effective refractive index. So, when measured using terahertz pulses, the porosity value is a bulk property of the entire tablet (that is, of the volumetric properties of the tablet). Therefore, these measurements can differ between samples, based on the samples’ thickness and refractive index.
Thicker samples will produce larger time differences (peak positions) in terahertz spectra. Differences in refractive index also affect the peak positions in terahertz spectra. Due to this, either the thickness or the refractive index must be known before the terahertz experiment is carried out. In the case of tablet measurements, the thickness should be known beforehand and used to later determine the refractive index. These calculations can vary based on the ingredients of the tablet, due to the fact that the calculations are dependent on the refractive indices of the materials being investigated.
What the data shows
The terahertz data acquired from the transmission spectroscopy measurements is Fourier transformed and compared to the data from a blank (empty sample chamber) data set. When comparing the sample spectrum to the blank, the largest peak will shift farther from the blank, depending on the thickness/porosity of the sample1. Figure 4 shows a representation of the peak shifts based on changes in the samples. Sample 1 (S1) is either thinner or more porous than Sample 2 (S2). Based on the distance between the sample peaks and the blank peak, and the known thickness, the effective refractive index of the sample can be calculated.
If the sample thickness is measured before the terahertz data is acquired and used to determine the effective refractive index, it can be used to identify porosity differences. So, as in the case illustrated in Figure 4, if the tablets have the same thickness, then the porosity of S1 is higher than that of S2. Using a calibration curve-like method, a set of data points can be collected and used to determine the measurements of unknown samples as well.
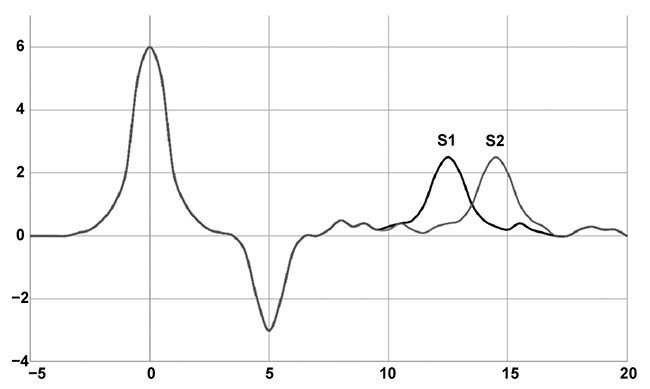
Figure 4. Representative terahertz spectra of a blank, Sample 1 (S1), and Sample 2 (S2). The graph shows the shift in first absorption based on tablet thickness or porosity differences. Courtesy of TeraView Ltd.
This ability is interesting because it
has the potential to predict disintegration
properties of tablets that have not undergone traditional disintegration tests. If a calibration curve can be built using THz-TDS measurements, then the disintegration properties for these products can be predicted. Although the industry may always use destructive methods of disintegration testing, the use of THz-TDS could provide a great deal of insight as to what results may be expected before those measurements are carried out.
The future of THz-TDS
Based on examples in research and in the manufacturing process, THz-TDS has been shown to be an effective new technique for measuring the porosity of pharmaceutical tablets. The ability of the method to enable quick and nondestructive evaluation of tablets has brought much benefit to the field, due to a reduction in the loss of valuable medicines. The ability to measure this quality enables insight into other important properties of tablets, especially dissolution and disintegration. Using THz-TDS to determine tablets’ effective refractive indices allows for the extraction of porosity data that is extremely useful for pharmaceutical researchers.
In the future, the information obtained from THz-TDS measurements of this key factor could be used to monitor the compaction processes of large-scale pharmaceutical production lines — for example, continuous manufacturing. Changes in procedures could be quickly detected and corrected. The technique could also provide added value to dissolution and disintegration evaluations because it can be used to predict these properties before destructive measures are used to determine their values8.
Meet the author
Thomas R. Juliano Jr., Ph.D., received a bachelor’s degree in chemistry from the University at Buffalo in 2011 and a doctorate in chemistry from Syracuse University in 2014. In May 2016, he joined TeraView Ltd. of Cambridge, England. He is currently the applications scientist and a member of the Applications Group for TeraView in the U.S..
Acknowledgments
This work was funded by Innovate UK, project reference No. 104196, and is a collaboration between the University of Cambridge, the
University of Strathclyde, TeraView Ltd., GlaxoSmithKline, and Huxley Bertram Engineering. The author acknowledges the work of Prince Bawuah, Daniel Markl, Daniel Farrell, Mike Evans, Alessia Portieri, Andrew Anderson, Daniel Goodwin, Ralph Lucas, and J. Axel Zeitler on the THz-TDS project.
References
1. D. Markl et al. (2017). Non-destructive determination of disintegration time and dissolution in immediate release tablets by terahertz transmission measurements. Pharm Res, Vol. 34, Issue 5, pp. 1012-1022.
2. D. Markl and J.A. Zeitler (2017). A review of disintegration mechanisms and measurement techniques. Pharm Res, Vol. 34, Issue 5, pp. 890-917.
3. Y.X. Bi et al. (1999). Evaluation of rapidly disintegrating tablets prepared by a direct compression method. Drug Dev Ind Pharm, Vol. 25, Issue 5, pp. 571-581.
4. W.A. Strickland et al. (1956). The physics of tablet compression XI: determination of porosity of tablet granulations. J Am Pharm Assoc Sci Ed, Vol. 45, No. 7, pp. 482-486.
5. P. Bawuah et al. (2014). Non-contact weight measurement of flat-faced pharmaceutical tablets using terahertz transmission pulse delay measurements. Int J Pharm, Vol. 476.
6. P. Bawuah et al. (2014). Detection of porosity of pharmaceutical compacts by terahertz radiation transmission and light reflection measurement techniques. Int J Pharm,
Vol. 465, Issue 1, pp. 70-76.
7. P. Bawuah et al. (2016). Noninvasive porosity measurement of biconvex tablets using terahertz pulses. Int J Pharm, Vol. 509, Issue 1, pp. 439-443.
8. P. Bawuah et al. (2020). Terahertz-based porosity measurement of pharmaceutical tablets: a tutorial. J Infrared Milli Terahz Waves, pp. 1-20.
/Buyers-Guide/TeraView-Ltd/c17011