
Thousands of Cells Tracked Via Lensless Imaging
LOS ANGELES, Sept. 18, 2012 — A novel lensless computational imaging platform is being touted as a new way to observe and track large numbers of rapidly moving objects under a microscope, capturing precise motion paths in three dimensions.
For the first time, the 3-D helical swimming patterns of more than 24,000 individual human sperm cells were directly recorded for as long as 20 seconds over wide fields of view and through large sample volumes. The work was done by a team at the UCLA Henry Samueli School of Engineering and Applied Science, led by associate professor of electrical engineering and bioengineering Aydogan Ozcan.
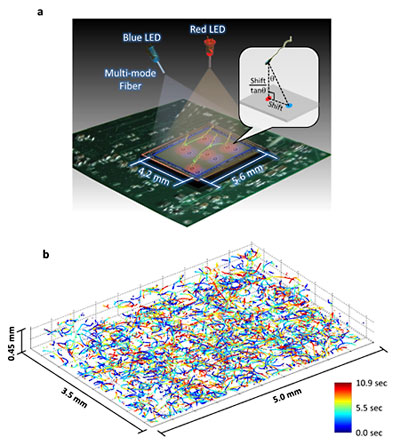
This illustration and data set depict the new microscopy technique developed by Ozcan and his colleagues at UCLA. The top image shows a schematic of the system, which involves two partially coherent light sources (a red 625-nm wavelength LED and a blue 470-nm wavelength LED) that simultaneously illuminate the microscope field of view from two different angles. A CMOS sensor records the resulting holograms, and software uses that information to encode the exact positions of the target cells. The bottom image shows the reconstructed 3-D trajectories of 1575 human male gamete cells in a volume of 7.9 microliters. (Images: Ozcan Research Group at UCLA)
"We can very precisely track the motion of small things, more than a thousand of them at the same time, in parallel," Ozcan said. "We were able to achieve submicron accuracy over a large volume, allowing us to understand, statistically, how thousands of objects move in different ways."
Observing human sperm cells has generally been limited to conventional lens-based optical microscopes. The small size of a sperm head (about 3 to 4 µm)requires a high-magnification lens to observe its motion, and sperm cells' relatively fast speed (about 20 to 100 µm per second) makes it difficult to track them over time as they move in three dimensions, into and out of the microscope's small observation field.
To address these challenges, the UCLA team developed a computational lens-free, on-chip imaging platform that uses the holographic shadows of sperm cells. Lens-free images of the cells were acquired simultaneously using two different wavelengths of light, one red and one blue. The offset light beams created holographic information that, when processed using sophisticated software, accurately revealed the paths of objects moving under a microcope.
This multi-angle, multicolor illumination setup allowed simultaneous and very accurate tracking of the 3-D motion of approximately 1500 individual human sperm cells (per experiment) across a wide field of view of 17 square millimeters.
The researchers found that human male gamete cells travel in a series of twists and turns along a constantly changing path that occasionally follows a tight helix — a spiral that, 90 percent of the time, is in a clockwise (right-hand) direction.
Because only 4 to 5 percent of the cells in a given sample travel in a helical path at any given time, researchers would have been unable to observe the rare behavior without the new high-throughput microscopy technique.
The research was published online Sept. 17 in Proceedings of the National Academy of Sciences. Other authors included UCLA graduate students Ting-Wei Su and Liang Xue, both members of the BioPhotonics Laboratory, which is directed by Ozcan.
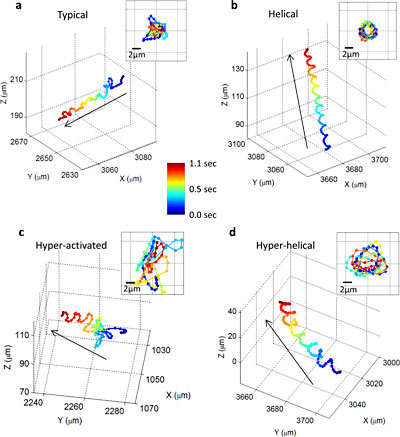
These images show, for the first time, that human male gamete cells swim primarily in four types of patterns. The insets show the front view of the trajectories.
"Our results demonstrate the unique capabilities of this high-throughput, on-chip computational imaging platform by resolving the tight and rapidly evolving, rare helical trajectories of motile human sperms, as well as by revealing their statistical behavior," Ozcan said. "This powerful lens-free imaging platform can also provide a high-throughput tool to rapidly quantify the impact of, for example, various stimuli, chemicals and drugs on the 3-D swimming patterns of sperms."
"The holographic technique could accelerate drug discovery and prove valuable for monitoring pharmaceutical treatments of dangerous microbial diseases," said Leon Esterowitz, the National Science Foundation (NSF) biophotonics program officer who has supported Ozcan's efforts.
This study is an extension of several years of NSF-supported work by Ozcan and his colleagues to develop lens-free, holographic microscopy techniques with applications for field-based detection of blood-borne diseases and other areas of telemedicine. Those efforts recently resulted in a Popular Mechanics Breakthrough Award and a National Geographic Emerging Explorer Award, among others. Ozcan's research is also supported through an NIH Director's New Innovator Award, Office of Naval Research Young Investigator Award and an Army Research Office Young Investigator Award from the Department of Defense.
For more information, visit: http://innovate.ee.ucla.edu or http://biogames.ee.ucla.edu
Published: September 2012