Laser Marking Turnkey Solution
TRUMPF SE + Co. KGRequest Info
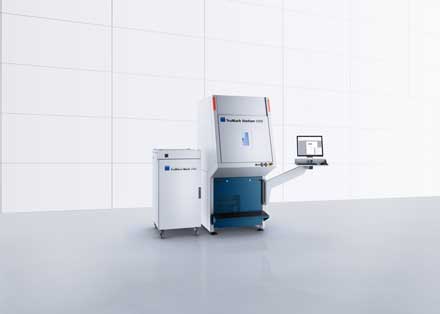 The TruMicro Mark 2000 turnkey solution for the Unique Device Identification (UDI)-compliant laser marking of medical devices has been announced by Trumpf GmbH, combining a marking laser with a UDI software module.
The TruMicro Mark 2000 turnkey solution for the Unique Device Identification (UDI)-compliant laser marking of medical devices has been announced by Trumpf GmbH, combining a marking laser with a UDI software module.
A single source, the device helps manufacturers comply with the current provisions set forth by the U.S. Food and Drug Administration that require every medical device to be labeled with a unique identifier that can be checked against a centralized database as part of the global device identification system.
The ultra-short pulsed laser features extremely short laser pulses of 0.4 to 20 ps and high pulse energies of up to 20 mJ. The UDI combines static and dynamic identifiers and is most often applied as a linear barcode or 2D data matrix code. The static portion is a unique code for the specific medical device while the dynamic portion changes for each batch of products.
https://www.trumpf.com/en_US
/Buyers_Guide/TRUMPF_SE_Co_KG/c15241
Published: April 2017
REQUEST INFO ABOUT THIS PRODUCT
* First Name:
* Last Name:
* Email Address:
* Company:
* Country:
Message:
When you click "Send Request", we will record and send your personal contact information to TRUMPF SE + Co. KG by email so they may respond directly. You also agree that Photonics Media may contact you with information related to this inquiry, and that you have read and accept our
Privacy Policy and
Terms and Conditions of Use.
Register or login to auto-populate this form:
Login
Register
* Required