
Green genes make glowing monkeys
For decades, scientists have bred genetically altered mice whose offspring have inherited their parents’ DNA modifications. For the first time, however, researchers have produced more than one generation of transgenic primates. Now it is hoped that the genetic engineering inroads that achieved this milestone will help breed primates with hard-to-treat neurodegenerative diseases such as Parkinson’s and amyotrophic lateral sclerosis, also known as Lou Gehrig’s disease, so that scientists can gain a better understanding of the human disease process.

Pictured on the cover of the May 28, 2009, issue of Nature is a marmoset named Kouichi, meaning “Kou’s first son.” He is the first second-generation transgenic primate. Courtesy of Nature.
As reported in the May 28, 2009, issue of Nature, scientists from seven Japanese institutions inserted the GFP gene into the DNA of embryos of the common marmoset Callithrix jacchus via a modified lentivirus, or slow-acting virus. GFP, which is isolated from the jellyfish Aequorea victoria, fluoresces green under blue and UV light and is used, for example, to track genes as they turn on and off, to illuminate tumor cells and to trace toxins.
The team, led by Dr. Erika Sasaki of the Central Institute for Experimental Animals in Kawasaki and by Hideyuki Okano of Keio University School of Medicine in Tokyo, injected 91 embryos with GFP, optimizing the process by placing them in a chemical soup that caused them to shrink. This allowed the void between each egg and membrane to be filled with more virus particles than was possible with earlier methods. The researchers then implanted a total of 80 embryos into 50 marmoset surrogates. Of the seven monkeys that became pregnant, three miscarried, but the remaining four produced five healthy babies, including twins Kei, a female, and Kou, a male. Their names combined form the word “keikou,” Japanese for “fluorescence.”
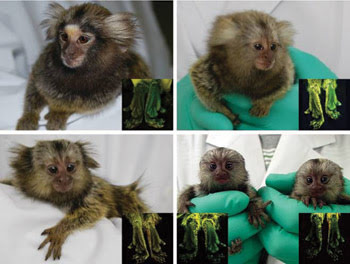
Shown are the five first-generation genetically modified marmosets. The two in the lower right are twins Kei (left) and Kou (right), named after the Japanese word “keikou,” which means “fluorescence.” The inset images show the animals’ green feet. Courtesy of Nature.
Although all five marmosets in the study are transgenic, only four express the transgene in their somatic tissues; i.e., four have skin and blood that glow green under UV and blue light. Besides a visual examination, the five primates were given a more sophisticated genetic test. The investigators confirmed that two – one female and one male – carry the GFP gene in their germ line (reproductive cells). The germ cells of the other three are still under analysis.
The first second-generation genetically modified marmoset, a healthy male billed as a “biomedical supermodel” on the cover of Nature (www.nature.com/nature/journal/v459/n7246), was the descendant of the transgenic male and a wild-type female. As for the three second-generation marmosets born since, two demonstrate the expression of GFP under a fluorescence microscope, but the third does not. The scientists are not sure whether this third marmoset expresses a level of GFP that is undetectable or whether the animal expresses no GFP at all; it has not been genetically analyzed.
The researchers chose the marmoset as an animal subject because it bears often and from a young age, and its reproductive cycle is more effectively controlled, enabling conception. The team is limited, however, by the constraints of the genetic engineering method it has developed. According to Okano, the size of the gene that can be introduced is limited to approximately 10,000 bases, or letters of the genetic code, so certain diseases – notably Huntington’s – most likely cannot be induced.
As for the possibility of imaging the neurons themselves, the first requirement, Sasaki said, is breeding a transgenic animal using a fluorescent protein expression cassette under a neuron-specific promoter; i.e., the DNA modification process must be more finely tuned.
Meanwhile, genetic engineering combined with fluorescence imaging should continue to advance the study of neuro-degenerative diseases.
Published: September 2009