... and also improves the manufacturing process.
Consumers are using the Internet to buy prescription drugs more cheaply, sometimes without a prescription, but they may get more than they bargained for because some unscrupulous retailers provide fake or adulterated drugs. The problem has been around for a long time, but the number of cases has increased with the growth of the Internet. Now, with the application of spectroscopy techniques new to the pharmaceutical industry, help is at hand.
According to facts compiled by the World Health Organization, counterfeit drugs account for more than 50 percent of those sold from Internet sites that conceal their physical address and, in developing countries in Africa, Latin America and Asia, from 10 to 30 percent of drugs sold are counterfeit. Criminals selling counterfeit drugs in these countries face few consequences.
In the US, legitimate pharmacies have bought drugs in bulk and sold them to less reputable wholesalers for a profit. The federal Prescription Drug Marketing Act of 1988 contains measures against this practice but leaves some major loopholes. For example, it fines hospital pharmacies that resell drugs to wholesalers but does not fine pharmacies based in nursing homes.
For drug manufacturers, this means that money is flowing out of their pockets and into those of illegitimate tradesmen, respected brand names can become diluted, and the manufacturer could be sued for selling a drug under false pretenses. The Center for Medicine in the Public Interest estimates that global sales of counterfeit drugs will reach $75 billion in 2010.
Patent protection?
Raman spectroscopy has become a standard method for identifying counterfeit drugs and also for FDA-required characterization of pharmaceuticals during the manufacturing process. The technique works because a laser induces molecules in a sample to vibrate, and these vibrations change the frequency of the light that is collected. Light with wavelengths close to that of the laser are filtered out. The resulting spectral profile is like a set of fingerprints that precisely identifies the chemicals in a drug or other substance.
Zai-Qing Wen, a scientist with the biotechnology corporation Amgen in Thousand Oaks, Calif., said that the company uses Raman spectroscopy daily. He stated that Raman spectroscopy has only recently become sensitive enough for analyzing protein pharmaceuticals.
Joe Hodkiewicz, director of Raman with Thermo Fisher Scientific, also said that Raman spectroscopy has become useful for industry only recently. He noted that near- and mid-infrared spectrometers, which Thermo also makes, have been used in the pharmaceutical industry for a long time. They work well for many applications, cost less and are more familiar to some scientists. However, Raman works best for some applications.
For example, Raman is useful for identifying the various crystalline forms of a drug, or polymorphs. These polymorphs not only potentially change the effect of a drug in the body, but also can affect patent rights. In 1997, the generic pharmaceutical manufacturer Novopharm won the right to make a generic version of the hugely popular drug Zantac because Glaxo’s patent on one polymorphic form ran out.
“Raman instruments were always aimed at an academic market,” Hodkiewicz said. As a consequence, experts in Raman spectroscopy could use them, but chemists with basic training in an industrial setting would have trouble with them. Additionally, they did not have the reproducibility needed for routine quality control in the manufacturing process. Thermo has focused on making Raman spectrometers easier to use and able to make reproducible measurements, as evinced by its DXR model.
Recently, Peter de Peinder and other scientists affiliated with the vibrational spectroscopy consultancy firm VibSpec in the Netherlands used Raman spectroscopy to detect counterfeits of the popular drugs Lipitor and Viagra, both manufactured by Pfizer Inc. “The advantage [of Raman] over other techniques is that it can be applied without sample preparation and sometimes even through the package,” de Peinder said. He noted that it also works nondestructively.
Pavel Matousek at the Rutherford Appleton Laboratory in the UK and others have developed spatially offset Raman spectroscopy, a variation of the classical technique that is particularly useful for identifying counterfeit drugs because it easily characterizes chemical compounds through opaque packaging. It also can detect drugs inside capsules during the manufacturing process.
Whereas textbooks teach to collect the Raman signal at the same region illuminated by the laser, practitioners of the spatially offset technique collect the signal beside the illuminated region. This works because fluorescence and the Raman signal from opaque containers obscure measurements made at the site of illumination. Matousek said that the classical technique is better for transparent containers, samples outside of containers and samples that do not fluoresce.
Because Raman spectroscopy is inherently a surface measurement technique, it can reveal the chemical components of a pharmaceutical coating with facility, but analyzing the core usually requires breaking the tablet in half. Even then, the method cannot easily detect the active pharmaceutical ingredient. Using polarized light can improve its ability to do so.
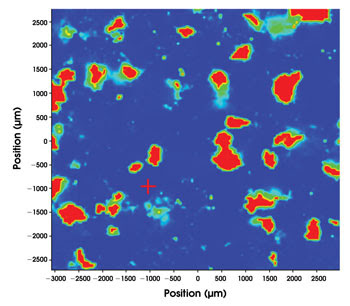
Raman imaging can provide a spectral profile that precisely identifies the chemical components of a drug. It has become a mainstay of pharmaceutical imaging.
“The difficulty with Raman is that you can’t image inside the tablet,” said Dan Arnone, CEO of TeraView Ltd., which is based in Cambridge, UK. On the contrary, terahertz radiation (t-ray) spectroscopy can show the chemical components inside a pharmaceutical tablet without requiring breaking the tablet in half.
Inside the pill
It also can show the release of the drug over time without the need for preparing a dissolution bath. Typically, a pill is dissolved in a bath with pH mimicking stomach acid, for example, to show how the drug would be released in the body, but terahertz can help drug makers design, predict and monitor drug release nondestructively.
Terahertz frequencies correspond to wavelengths from the microwaves to the end of the infrared spectrum. The company achieves terahertz wavelengths using femtosecond lasers to irradiate semiconductor materials, particularly gallium arsenide. Its tablet imager co-developed with Pfizer, the TPI imaga 2000, uses a robotic arm to pick up a tablet from a tray of tablets and rotate it through a terahertz beam.
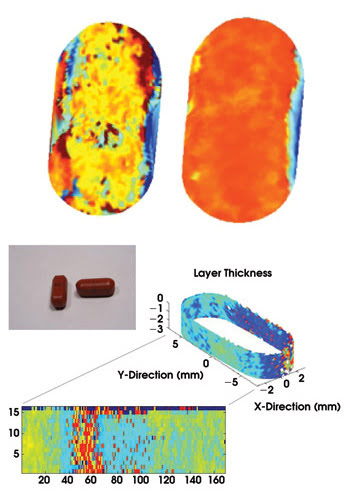
Terahertz radiation can image inside a pill without requiring that it be destroyed or broken in half.
It should be noted that t-rays work well for characterizing drugs and other chemicals in solid form, such as tablets, but have trouble passing through liquids for characterizing liquid dosage forms.
Arnone said that the main challenge to acceptance of terahertz technology has been the conservative nature and high regulation of the industry, but that the technology will be an asset once accepted. “We have been working with the FDA for four years, and now we’re at a stage at which we’re working with customers trying to get a drug through the approval process,” he said.