Lynn Savage, Features Editor, lynn.savage@laurin .com
So much of what needs to be learned about human and animal health, evolution, environmental science and a myriad of other topics begins and ends with cell biology. Not so long ago, researchers could look at only the most basic results when studying cell behavior: What happens if I expose a group of cells to a particular chemical, to a virus or to ultraviolet light?
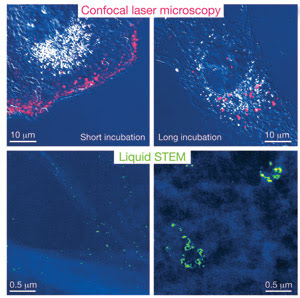
The images on top were acquired using standard confocal microscopy. The images on the bottom were obtained using a technique called “liquid STEM,” in which the cell is placed in a liquid environment within a microfluidic chamber, then imaged with scanning transmission microscopy. The researchers report that the resolution is about 50 times better with liquid STEM than with confocal microscopy. Images courtesy of Niels de Jonge, Vanderbilt University Medical Center.
Microscopy is the cell biologist’s best friend, and techniques continue to improve – in some cases, surpassing the diffraction limit that restricts resolution to the wavelength of the light used to image a cell. Currently, the best resolution is 10 nm – very good, but it comes at the cost of slow imaging times. Fast processes, such as the migration of various cellular components and their interactions with one another, must be imaged with fast frame rates, or else information will be lost. You can have high resolution or high speed, but not both, unless you freeze a cell, place it in a vacuum and image it with electron-beam or x-ray diffraction techniques, but then the cell is out of its native liquid surroundings and frozen in place.
Now researchers at Vanderbilt University Medical Center and Oak Ridge National Laboratory, led by Niels de Jonge of the department of molecular physiology and biophysics, have developed a way to get around the limitations of imaging in a vacuum.
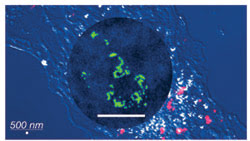
In this composite, images acquired using a confocal laser microscope are overlaid by an image acquired using the liquid STEM method (in circle). The scale bar in both cases is 500 nm. The labels are individually visible with liquid STEM. The confocal image was obtained from a different sample (in which the labels are quantum dots) than that used for the liquid STEM image (where the labels are gold nanoparticles). The round shape is not the cell nucleus.
As described in a forthcoming issue of Proceedings of the National Academy of Sciences, de Jonge’s group developed a microfluidic chamber in which eukaryotic cells in liquid were covered by “ultrathin, electron-transparent” windows. The apertures permitted them to bring the samples into a high-vacuum chamber and to perform scanning transmission electron microscopy (STEM) on them.
The technique, dubbed “liquid STEM,” provides a resolution of 4 nm at a pixel dwell time of 20 μs. A 1024 × 1024-pixel image of a cell takes 21 s.
De Jonge said liquid STEM should aid in the creation of better molecular probes and lead to a better understanding of interactions between cells and viruses.