David L. Drapcho and Kris Ver Donck, Digilab Inc., with Terry McCann, TJM Consultancy
High-content screening is the collection of multiple data parameters in parallel from the same cellular samples. It has evolved into a mature technique typically using automated fluorescence microscopy and high-resolution imaging of cells stained with multiple labels. Very little effort has been expended on analyzing bright-field images because obtaining accurate statistical data from bright-field images alone has been considered too difficult.
But bright-field image analysis enables the study of unlabeled live cells in their natural state, unperturbed and undamaged by chemical modification. Collecting multiple images of the same cells, including time-lapse experiments, can be a powerful tool for understanding the effect of stimuli on cell processes.
The MIAS-2 imaging system
The MIAS-2 from Digilab Inc. enables collection of bright-field and fluorescent images with automated analysis in both acquisition modes. The reader, schematically shown in Figure 1, is based on inverted epifluorescent microscopy optics that can collect high-resolution images in multiple bright-field and fluorescent channels.
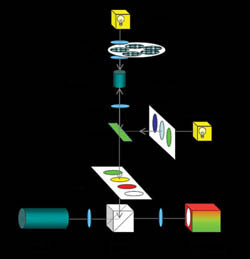
Figure 1. In the MIAS-2 imaging system, bright-field images are obtained in transillumination configuration with a 100-W halogen source, and fluorescent images are recorded in an epifluorescent configuration with a 70-W high-pressure xenon source. Bright-field and fluorescent images are collected on an EMCCD camera, with an option for image collection with a color CCD camera.
The MIAS-2 uses a 14-bit electron-multiplied (EM) CCD camera for collection of images in both modes, allowing for direct overlay of images without software correction, since both image sets are collected from the same optical path. In addition, the camera enables imaging of weak fluorescent samples, shortening acquisition time in some cases by orders of magnitude. To provide flexibility in sampling a large variety of cell types, the MIAS-2 reader is equipped with motorized objectives ranging from 2.5× to 63× with high numerical apertures. An object-based autofocus algorithm offers accurate and reproducible focusing.
The MIAS-2’s enabling technology for analysis of bright-field images is not in the hardware, because any high-quality microscope can collect high-resolution bright-field images. Instead, the technology is in the proprietary image analysis algorithms developed by Digilab. Examples of these algorithms follow.
Bright-field applications
Some benefits of bright-field image analysis are as follows:
• Avoidance of the effects of chemical labels or genetic modification, which can alter cellular behavior
• Avoidance of the time and cost of complicated chemical staining procedures, which may be difficult to automate
• Option of repeated observations of the same cell cultures over time
• Little to no phototoxicity with images collected at low light levels
• Image-based quality control of cells before and after biochemical assays
In short, label-free imaging avoids the disadvantages inherent in fluorescent probes, thereby sidestepping the cost and complexity of preparing fluorescent assays while providing much of the same information. Label-free applications available for the MIAS-2 reader in bright-field mode include cell-based assays such as cell count, clonality, colony size, cell migration, cell-cell adhesion and neurite outgrowth (See Figure 2). Animal-based assays for nematode embryonic development, toxicology and behavior also are offered.
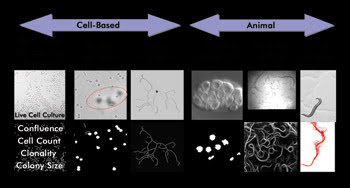
Figure 2. Label-free applications available for the MIAS-2 reader in bright-field mode include cell- and animal-based assays. The bright-field image is displayed above the digital (analysis) image.
Obtaining high-quality images of nonlabeled cells in bright-field mode is challenging, but analyzing these images is perhaps even more so. The incorporation of scale space mathematics in image analysis algorithms allows the identification of objects based on the mathematical description of their shape rather than on an intensity above a threshold. This feature circumvents many of the problems imposed by background variations that are so typical of bright-field images.
The advantage of this technique is obvious in rapidly evolving cell culture-based research. In the past, the number of cell lines used was limited, and often the same transformed cells were used by most researchers. Today, cell culture techniques have advanced, and scientists use many different cell types, including primaries and stem cells. All have particular morphological properties, and the imaging software must be able to handle all possible variations and combinations of cell types. In this regard, it is essential to develop tunable applications, where the user can define a number of basic parameters without detailed knowledge of software programming. Tunable applications may not be sufficient to tackle all possible variations, however, so the development of custom-made applications remains an option. To ensure maximum flexibility in image analysis, an open data structure with standard image file types (tiff and jpeg2000) allows images to be imported into, or exported from, the reader software.

Figure 3. Results of the confluence analysis to the measurement of cell growth of the same cell culture over four days are shown with the bright-field image (top row) and with the confluence analysis result superimposed in false color over the bright-field image (bottom row).
One benefit of label-free live-cell imaging is that it allows repeated observation of the same cell culture over multiple days using a confluence analysis. Figure 3 shows the results of the confluence analysis to the measurement of cell growth of the same cell culture over four days. Adherent cells were seeded into the well, measured on the MIAS-2 reader for confluence and placed in an incubator. The plates were taken from the incubator once per day to be re-imaged in the MIAS-2 reader, then placed back in the incubator for continued growth. This process was repeated over four days. For convenience, a climate control option is available for the MIAS that allows a single plate to be continually kept in the reader over the incubation time. This application replaces a fluorescent measurement using a cellular stain, which in most cases alters cellular growth and has to be performed on various cell cultures.
Another common fluorescent application is staining cell nuclei to perform a cell count, but this analysis can be performed just as well on a bright-field image using a biochemical assay.

Figure 4. Results for a control and for nocodazole-treated cells at 100-nM and 1-nM concentration are shown left to right, with the bright-field images (top) shown over the image analysis results superimposed in false color over the bright-field image (bottom).
Figure 4 shows the application of cell count analysis for measurement of growth inhibition of primary human keratinocyte cells. Cells were seeded into the wells of a microtiter plate, treated with different compounds at varying concentrations, incubated, then measured after 24 hours for cell count. The results demonstrate little inhibition of growth for the cells treated with 1 nM nocodazole, whereas a treatment at 100× higher concentration clearly prevents cell growth. On the right is shown a comparison of an MTT biochemical assay to IC-50 results for nocodazole and a number of other compounds obtained by bright-field imaging on the MIAS-2, demonstrating a direct correlation between the two sets of results.
As the data in Figure 4 demonstrate, the bright-field analysis of unlabeled live cells works just as effectively as a sensitive assay of biochemical activity.
Fluorescent applications
Several assays, however, still require fluorescent stains and a sensitive system to collect and analyze the images. One such assay is exemplified in Figure 5, for a nuclear assembly of GFP-labeled lamin. Lamin is a low-abundance protein involved in the assembly of the nuclear membrane that disassembles during cell division. This assay takes full advantage of the EMCCD camera in the MIAS-2 reader, where a 30-ms integration time replaces a 10- to 30-s measurement on a normal CCD, decreasing the measurement time by two to three orders of magnitude.
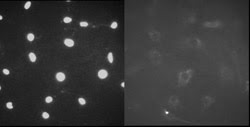
Figure 5. Nuclei can be visualized in the image on the left, but the lamin disperses into the cell cytoplasm during cell division (right). The fluorescent signals in these images were too weak to be seen by the human eye and could be visualized in the EMCCD camera only by increasing the gain.
The MIAS-2 reader is available with a number of common applications based on fluorescent images, such as assays for nuclear and cytoplasmic stains, apoptosis, nuclear condensation, receptor clustering, micronucleus count and others for cell-based assays, and vascular structure and pharynx pumping for animals. Thus, the field of fluorescent imaging is not neglected by the MIAS-2 reader but enhanced by the development of the bright-field applications.
Not all applications in high-content screening can be performed on bright-field images, so it is a distinct advantage to have a system that can perform both analyses: on the bright-field images where possible, and on fluorescent images where specific subcellular features must be imaged.
Meet the authors
David L. Drapcho, PhD, is general manager of marketing at Digilab Inc. in Holliston, Mass.; e-mail: [email protected]. Kris Ver Donck is Digilab’s chief technical officer, imaging, in Geel, Belgium; e-mail: [email protected]. Terry McCann is an independent business consultant to the life sciences industry based in Sevenoaks, UK; e-mail: [email protected].