
Technique probes blood cell membranes
Researchers have employed diffraction phase microscopy to gain more
information about the morphology – form and structure – of human red
blood cells. Their investigation could lead to advances in the treatment and screening
of blood-cell morphology diseases such as malaria and sickle cell. The doughnut-shaped
cells contain the molecule hemoglobin, which carries oxygen from the lungs throughout
the body. They make up 40 to 45 percent of a person’s blood. The cells, which
have indented centers, lack the internal structures found in other cells.
Red blood cells have a membrane structure that enables them to
be flexible and resilient, allowing them to squeeze through capillaries half their
diameter. The cells transform from normal to spiculated to nearly spherical shape
while accompanied by changes in cell mechanics. Little has been known about the
mechanics of this membrane, a fluid lipid bilayer with an elastic 2-D network of
spectrin, a cytoskeletal protein.
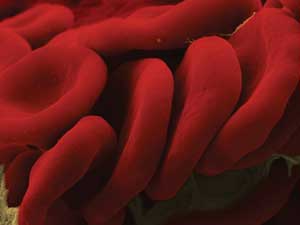
Researchers used diffraction phase microscopy to study the mechanics of human red blood cell
membranes. The cells are shown here. Courtesy of Gabriel Popescu.
Diseases such as malaria, spherocytosis and sickle cell can cause
changes in both the equilibrium shape and mechanics of the cells, which affect their
transport function, according to the scientific report by YongKeun Park et al.,
published online in PNAS on March 29, 2010.
The interdisciplinary team, led by professors Gabriel Popescu
(electrical and computer engineering at the University of Illinois at Urbana-Champaign)
and Alex Levine (chemistry and biochemistry at the University of California, Los
Angeles), used diffraction phase microscopy to quantify the undulations of the membranes
during the shape transition. The team was interested in learning how the deformability
(alteration of form or shape) of the red blood cells related to their morphology.
The deformability of the cells is considered to be their most important property.
The highly sensitive measurement technique uses two beams of light
– one going through the specimen and the other used as a reference. It enabled
the scientists to see nanoscale membrane fluctuations in live cells and to measure
them quantitatively. The researchers applied the noncontact optical interferometric
technique to quantify the thermal fluctuations of the cell membranes with 3-nm accuracy
over a range of spatial and temporal frequencies. By measuring “bumpy”
red blood cells called echinocytes and round ones called spherocytes, they discovered
that these deformed cells display less flexibility in their membranes – a
finding that could provide insight into the mechanics and treatment of the diseases
that affect the shape of red blood cells.
The researchers used a new mathematical model of the red blood
cell membrane fluctuations to extract from their fluctuation data the elastic properties
of the red blood cell membranes. This mathematical model accounts for the curvature
of the cells. Previous models had treated the membrane as a flat sheet.
The dual optical method and theoretical model allowed them to
find some new results where the shape and deformability are coupled.
Microrheology
Levine, who was involved in the theoretical aspects of the research,
explained that the basic framework within which this sort of modeling falls is called
microrheology. He said the basic idea is that in the macroscopic world it is fairly
straightforward to push – or, more technically, shear – materials to
measure how they deform or flow under stress, which is the study of rheology.
“When you wish to study the rheology of microscopic and
very fragile materials, it is obviously difficult to grab them and push on them.
This is where the idea of studying the thermally driven fluctuations of soft microscopic
materials comes in. By observing their fluctuations, you can work backwards mathematically
to determine their elastic/mechanical properties,” Levine stated.
“Popescu’s experiments provide a beautifully precise
measure of the fluctuations of the membranes of these cells. That membrane is only
nanometers thick and a few microns across. It is both extremely tiny and fragile
on the scale of typical macroscopic objects. Our mathematical models allow us to
analyze that fluctuation data and determine in detail the various elastic properties
of the membrane. So this is an excellent example of microrheology being applied
to cell membranes,” Levine said.
The dual optical technique and mathematical model could have applications
such as screening for certain blood diseases, and screening stored blood for membrane
flexibility prior to transfusion – cellular changes in banked blood often
occur. It may also allow scientists to study the effects of chemical agents, such
as alcohol, on membranes, and to assess the effectiveness of medications in development
for the treatment of blood cell morphology diseases.
The investigation included collaborators from Massachusetts Institute
of Technology in Cambridge, Mass.; from Harvard Medical School, Harvard School of
Engineering and Applied Sciences, and Massachusetts General Hospital, all in Boston;
and from the University of Colorado at Boulder.
Published: September 2010