By David L. Carroll1 and Claudia K. Gunsch2
1Center for Nanotechnology and Molecular Materials and Department of Physics
Wake Forest University
Winston-Salem, N.C.
2Center for the Environmental Implications of Nanotechnology and Department of Civil and Environmental Engineering,
Duke University
Durham, N.C.

Nanoparticles released into the environment can have a wide range of biological effects. These effects can depend not only on the specific chemical makeup of the nanoparticles in question, but also in the aggregate morphology that the materials may take as they move through a given ecosystem. There is growing awareness of the impacts that free and aggregate forms of nanomaterials may represent and the research directions that are necessary to mitigate the potential risk of these materials.
With the continued explosion of opportunities in industrial applications of nanomaterials and their anticipated pervasiveness in near future marketplaces, there has been increasing interest in the environmental impact of such materials. While it is impossible to discuss in detail all of the ongoing activities pertaining to the environmental impacts of nanomaterials here, it is clear that researchers are beginning to coalesce around a few key themes. The main research topics include: 1) the methods and morphology of release of nanoparticles into natural environments, 2) the corresponding toxicology of those materials, 3) the movement and persistence of nanomaterials within the environment, and finally 4) the ultimate fate and sequestration of nanomaterials in the environment.
What we have learned so far is that toxicological interactions between nanomaterials (nanoparticles) and life forms in the environment can take several routes. Specific toxicity to an organism (particularly at the cellular level) is typically thought of in terms of the effects of biotic uptake where chemical interactions or free radical production may cause extensive damage. Uptake of nanoparticles can depend on a variety of factors such as the size and shape of the nanoparticles that interact with the organism or the surface ligands on the nanoparticle which alters surface charge as well as the type of organism. Cellular endocytosis for instance typically requires some form of acceptor activation implying that the nanoparticles’ chemistry is extremely important for this process to occur. However, direct membrane penetration has been shown to occur for those particles which are extremely hydrophobic such as nanotubes, and fullerenes, as long as their overall diameter is smaller than about 20 nm. Cellular entry through transmembrane channels is usually only seen for nanoparticles smaller than 5 nm in diameter. Cellular uptake can enhance overall bioaccumulation of nanoparticles in an organism leading to questions of critical dosing.
However it is important to remember that nanoparticles in the environment, particularly if their route of exposure is waterborne, are likely to have previously interacted with other materials in the environment. Since many large molecules such as free proteins act as surfactants for nanoparticles, free nanoparticles in a variety of environments (e.g., sea water) can quickly adsorb a layer of such material. With the nanoparticle’s ability to transfect cellular structures, it becomes possible for such materials to be transported within cells leading to a facilitated toxicity.1 This facilitation of toxic effects is well demonstrated not only by adsorbed chemical species that may be brought into cells, but also by the reactions that may be driven by external stimuli found in the environment. An example of this is the use of TiO2 (titania) nanoparticles in the presence of UV radiation.2 Titania is an extraordinarily good photocatalyst and, in the lab, the exposure to such radiation is not tremendous. However, in the environment, UV catalysis can lead to free radical production and the formation of acidic species that can be harmful to life forms. This is also true for nanoparticles of metals which may be released into sea water.3,4
In terms of human health, the problem of nanoparticle driven complement activation5 also falls within the rubric of “facilitated toxicity.” Nucleophilic adsorbates on the surface of the nanoparticle can provide alternative pathways to the generation of anaphylactic agents such as C3A and C5A, leading to the generation of leukocyte receptors C3BC5B. This in turn can form membrane attack complexes (MACs) leading to tissue damage. Alternatively, antibody recognition of denatured autologous proteins that were adsorbed to the nanoparticles may yield cross-reactions with “native” proteins, inducing auto-immunity.
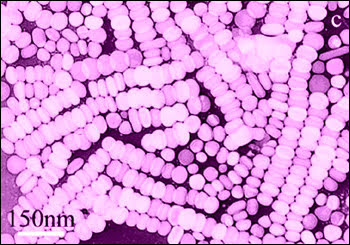
Transmission electron micrograph of CTAB stabilized Ag nanoparticles made from AgNO3 Ascorbic Acid and NaOH.6 (Image: David L. Carroll)
A common theme in environmental toxicology of nanoparticles is that the form that such materials take in nature is generally aggregated, so individual nanoparticles are rarely found interacting with a life form. While there are no definitive studies at this point as to the morphology of persistent nanomaterials in the environment, certainly aggregates can be found in aerosols and aqueous suspensions of nanoparticles. In many ways, these morphological forms may be of more concern.7 Morphological toxicity can be derived from occlusions in biological organisms which can be inhibiting to physiological processes. In the case of airborne risk, aggregates of titania,8 carbon nanoparticles9 for example, that are smaller than one micron have been shown to be severely detrimental to lung tissue and airway passages causing embolism. Furthermore, while granuloma formation may result in this case, yielding little toxicological impact, clearly reduction in function from extensive exposure could result. A similar example has been seen in vascular occlusion due to over exposure to carbon nanotubes in the blood stream.10 Finally, the ability of such aggregates to shed nanoparticles, once within an organism, has not been well studied and represents another potential risk.
These toxicological effects are obviously not the only impacts that might be anticipated. What is now coming into focus is that an impact may not need to be presented by toxicity to a specific organism. We have already learned many lessons from the introduction of chemicals into our environment, such as pesticides, where no specific toxicity to animal life was anticipated, yet endocrine disruption (for instance) turn out to be just as deadly to the species. So, as these nanomaterials integrate into the larger macrocycles of our ecology, specific synergistic processes may be at risk as well, leading to propagation of harm to organisms throughout the system. At this point, these factors are quite simply unknown.
The challenge in determining the overall long term impact of nanomaterials is to understand the dynamic process in terms of the changing nature of the nanoparticle’s toxicity, their environmental persistence and their facilitation of other toxic routes. This makes it difficult, if not impossible, to propose a single model to encompass the many families of nanoparticles. Thus, the taxonomy of any given family’s environmental and biological interactions must be taken independently of others. That is carbon nanotubes must be treated differently from boron nitride nanotubes for instance, and one principle may not hold for both when it comes to cellular uptake and/or toxicity with a given ligand.
The challenge for the photonics community is the design and implementation of highly sensitive optical tools for the detection of both the presence and the state of nanomaterials in the environment. Standoff optical techniques can take advantage of numerous optical signatures, but they tend to be materials dependent. Single walled carbon nanotubes, and nanoparticles of semiconductors are highly luminescent, whereas metal nanoparticles are among the best chromophores known. Undoubtedly, the most successful approaches will involve coupling luminescence signals with nonlinear processes such as Raman signatures (for example) as a complimentary way of extracting the state of the materials in the environment. It should be recognized however that this field is very much in its infancy and there is much room for innovation.
The good news is that the scientific community, together with funding agencies such as the NIH, NSF and EPA, has been aggressively proactive in understanding these risks. While there is still much to be done, Programs at Universities such as Wake Forest University and research centers such as the Center for the Environmental Implications of Nanotechnology at Duke University along with many others across the U.S., Europe and Asia, continue to lead the way in developing meaningful assessments as to the impact these materials may have on our environment and ultimately human health.
The future of nanoparticles in commercial processes and products does indeed hold much promise. And, perhaps they represent no more risk than any other class of new materials (such as polymers) have done in the past. Still, they do present different challenges for the scientific community. However, ongoing efforts to understand these materials in advance of their widespread utilization are perhaps the first instance of the scientific community’s ability to be proactive in the introduction of a new material to our environment. We are likely to understand the risks of these materials, far better than in any other example in history, before they become widely available.
________________________________________________________________________________________________________________
1Nicole Levi, Roy R Hantgan, Mark O Lively, David L Carroll, Gaddamanugu L Prasad, C60-Fullerenes: detection of intracellular photoluminescence and lack of cytotoxic effects, Journal of Nanobiotechnology 2006, 4:14 (14 December 2006)
2Yuanzhi Li, Nam-Hee Lee, Jae Sung Song, Eun Gu Lee and Sun-Jae Kim, Synthesis and photocatalytic properties of nano bi-crystalline titania of anatase and brookite by hydrolyzing TiOCl2 aqueous solution at low temperatures, Research on Chemical Intermediates Volume 31, Numbers 4-6, 309-318, DOI: 10.1163/1568567053956554
3Amy H. Ringwood, Nicole Levi-Polychenko, David L. Carroll, Fullerene Exposures with Oysters: Embryonic, Adult, and Cellular Responses, ENVIRONMENTAL SCIENCE & TECHNOLOGY Volume: 43 Issue: 18 Pages: 7136-7141 Published: SEP 15 2009
4Amy H. Ringwood, Melissa McCarthy, David Carroll, The Effects of Silver Nanoparticles on Oyster Embryos, Marine Environmental Research, doi:10.1016/j.marenvres.2009.10.011 (2009)
5Sai T Reddy, André J van der Vlies, Eleonora Simeoni, Veronique Angeli, Gwendalyn J Randolph, Conlin P O'Neil, Leslie K Lee, Melody A Swartz & Jeffrey A Hubbell, Exploiting lymphatic transport and complement activation in nanoparticle vaccines Nature Biotechnology 25, 1159 - 1164 (2007)| doi:10.1038/nbt1332
6Sihai Chen, and David L. Carroll, “Silver Nanoplates: Size Control in Two Dimensions and Formation Mechanisms,” J. Phys. Chem. B 2004, 108, 5500-5506
7Junko Okuda-Shimazaki, Saiko Takaku, Koki Kanehira, Shunji Sonezaki, and Akiyohshi Taniguchi, Effects of Titanium Dioxide Nanoparticle Aggregate Size on Gene Expression, Int J Mol Sci. 2010; 11(6): 2383–2392. doi: 10.3390/ijms1106238.
8Timothy R Nurkiewicz, Dale W Porter, Ann F Hubbs, Jared L Cumpston, Bean T Chen, David G Frazer and Vincent Castranova, Nanoparticle inhalation augments particle-dependent systemic microvascular dysfunction, Particle and Fibre Toxicology 2008, 5:1doi:10.1186/1743-8977-5-1
9Leah A. Mitchell, Jun Gao, Randy Vander Wal, Andrew Gigliotti, Scott W. Burchiel Jacob D. McDonald, Pulmonary and Systemic Immune Response to Inhaled Multiwalled Carbon Nanotubes, Toxicological Sciences, Volume100, Issue1, Pp. 203-214.
10Anna Radomski, Paul Jurasz, David Alonso-Escolano, Magdalena Drews, Maria Morandi, Tadeusz Malinski, Marek W Radomski, Nanoparticle-induced platelet aggregation and vascular thrombosis, British Journal of Pharmacology, Volume 146, Issue 6, pages 882–893, November 2005