Barbara Foster, The Microscopy & Imaging Place Inc.
Research biology is changing, moving toward more complex experiments that combine imaging and traditional fluorescence with photostimulation and electrophysiology. As these new directions evolve, they require light sources that adapt on the fly, rapidly providing multiple wavelengths in a format that is programmable and tunable.
Nature has been using light to stimulate action for eons. Light-sensitive proteins activate phototropism (growing toward the light), photoperioidism (timing flowering based on length of day and night) and photoplasty (seed germination) as well as circadian rhythm and sensing of magnetic fields. Since the mid-1980s, microscopists have been light-activating fluorophores, first just to locate structures of interest, and then – with the expansion of Fura-2 and similar probes – to track processes such as metabolism and cell signaling.
The next evolutionary step now emerging is optogenetics: using light to control behavior at the cell membrane, across the membrane into the cytoplasm, across groups of cells acting as sensor arrays, or within a human or living animal as a whole. It has been developing so dramatically, it was named Method of the Year by Nature Methods in 2010.1,2
Today’s innovative light sources can be used for a broad range of experiments. More traditional research might use FURA-2 to study Ca++ behavior in T-cell activation as part of the immune response. Cutting-edge optogenetics studies might combine imaging with photoactivation and electrophysiology.
One such light source is the Lambda DG-4/DG-5 Plus from Sutter Instrument Co. The source has dual galvanometers (Figure 1): The first directs light from a 300-W xenon lamp into the light path; the second collects the light and redirects it into the lightguide, which acts as the connection point between the illuminator and the microscope or other experimental setup. The filter holder arrives at the customer site empty, allowing the user to insert the filter set that best fits each experimental design. For example, for a FURA-2 experiment, a microscopist might choose narrowband interference filters for 340 and 380 nm, leaving a third position available as a white-light position. Conversely, an optogenetics experiment might require one specific wavelength, such as 470 nm, pulsed at specific time intervals, as described below in the experiment using channelrhodopsin-2.
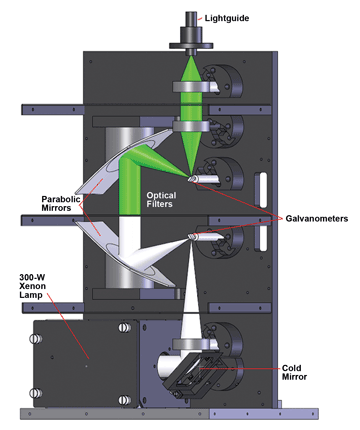
Figure 1. The Lambda DG-4/DG-5 Plus optimizes the light path and provides tunability using parabolic mirrors and scanning galvanometers. Courtesy of Sutter Instrument Co.
Digitally controlled galvanometers deliver very fast switching between filter settings without any of the vibration typical with a moving filter wheel. Typical on/off time is 500 μs, suitable for following fast changes in ion concentrations and many neuronal processes as well as for real-time video. To ensure correct switching time, the galvos tune themselves automatically. Moving the input and output galvos out of sync when traveling between nonadjacent filter positions also provides a turbo-blanking feature, preventing the sample from being exposed to light at unwanted wavelengths.
Two parabolic mirrors increase the efficiency of light moving through the system. The first sits immediately after the entry galvanometer to collect and collimate light onto the interference filter of choice. Its partner is positioned
behind the interference filter, capturing the emerging light and focusing it onto the exit galvanometer.
Finally, using a liquid lightguide as a connector scrambles the light as it exits to the microscope, providing a uniform field of illumination. The cold mirror placed just after the light source eliminates IR radiation, extending the lifetime of the optics and the lightguide, and preventing exposure of the sample to IR in the white-light setting.
Flexibility, tunability, programmability
Such a flexible light path provides a number of advantages for both optogenetics and general biological research. Capitalizing on modern interference filters optimizes both wavelength selection and throughput.3 Other narrow-bandpass systems such as single-cavity interference filters, grating monochromators and acousto-optical modulators often pass unwanted harmonics, and variable-wavelength devices cannot always sufficiently block out-of-band wavelengths. Allowing the user to choose the filters enables an easy transition from conventional bright-field microscopy to an infinite number of modes for research biology or optogenetics. Further, since the filters can be readily exchanged, it is ideal for a multiuser facility.
A 300-W xenon lamp provides a continuous spectrum from the near-UV through the near-IR, enabling researchers to take advantage of the full range of evolving optogenetic probes. Although LEDs are capable of faster switching, wavelengths currently available do not always match the necessary dyes and optogenetic probes.
Intentionally misaligning the exit galvo also provides neutral density filtration. Intensity can be attenuated down to 30 percent with good accuracy and repeatability. For greater attenuation, the user can insert conventional neutral density filters.
The instrument is fully programmable, enabling the user to build a sequence and step through it. Two different outputs offer connectivity to either a computer or a data recorder. The modes of operation can be selected manually using a keypad or via a computer. The device offers 16 programmable galvo/filter setting positions and can also be reprogrammed to custom settings; e.g., one wavelength at several different intensity levels, two other filters at 100 percent, and the last filter attenuated. A ring buffer allows the user to set a sequence, then step through it using a TTL (transistor-transistor logic) trigger pulse.
Determining calcium’s role in cilial beating
Early in his career, Dr. Richard J. Bookman of the University of Miami Miller School of Medicine recognized the role of Ca++ in modifying cell function as a unifying biological concept. A key focus of his work involved measuring intercellular calcium in nerve cells. Calcium plays a critical role in the release of neurotransmitters, fundamental to the process of synaptic communication. However, in researching the impact of calcium on nerve function, Bookman’s group ran into several challenges. For one thing, calcium fluxes are rapid, requiring high temporal resolution. For another, imaging the structures themselves requires high spatial resolution.
These diametric needs dictated protocols requiring two very different and complex imaging systems: one that was PMT-based to spatially integrate from the full field, providing high spatial resolution, and a second that was iCCD-based for video rate imaging. However, the CCD requires time to acquire the image on sensor, then time to read out. A specific wavelength must be kept in place for a finite period, then rapidly switched (at a submillisecond rate) to another wavelength. As is typical in an academic setting, Bookman’s group had more human capital than financial resources, so the decision was made to invent what they needed – and the DG-4 was born.
The role of calcium in effecting cilial beating is a case in point. To conduct this experiment, the researchers patch-clamped a single cilium from a single cultured tracheal epithelial cell. They determined the beating frequency of the cilium (3, 5, 10, 12 Hz) by conducting a Fourier transform on the transmitted light signal from the phase-contrast image. Simultaneously, to determine the mechanism and the role that calcium plays in controlling the frequency of ciliary beating, they conducted ratiometric imaging using Fura-2 to measure the level of intracellular calcium. The combination of techniques yielded the kinetics of the process and enabled them to put upper and lower bounds on the calcium concentrations involved.
Uncaging to catch T-cell motility
Dr. Peter Beemiller works in a multiuser facility at UCSF, where he finds it advantageous to pop filters in and out, rapidly converting from standard bright field or fluorescence to his own dedicated protocols. His work centers around T-cell activation, an important part of the immune response. Activation occurs when T-cell antigen receptors (TCRs) bind with antigen-presenting cells (APRs), forming an immunological synapse.4,5
For their uncaging experiments, Beemiller’s group uses UV illumination, connecting the light source to a mosaic, which allows them to illuminate selected sections of the field, avoid protected areas and hit only targeted molecules. For his animal model, he prefers using transgenic mice with T-cells modified to recognize albumin because no pathogens are involved. To image, his group uses time-lapsed TIRF (total internal reflection fluorescence), placing the living cell lipid bilayer directly on a glass coverslip. This protocol simplifies the microscopy. Because all the events are happening right on the glass, concerns about cell orientation and movement are eliminated. Changing wavelengths faster than a filter wheel is important for capturing T-cell motility, Beemiller said.
Optogenetics: Combining imaging with electrophysiology
Optogenetics uses light to stimulate behavior and genetics to introduce or express the necessary light-responsive proteins into the cells or organs of interest.6 Unlike deep-brain electrical stimulation, which electrically affects a general area of the brain, optogenetics targets very specific processes. Depending on the protein used, the light either activates or silences that process. As discussed in the classical article by Dr. Feng Zhang of MIT and his then colleagues at Stanford University,7 optogenetics can be applied to “electrically excitable cells [including] skeletal, cardiac and smooth muscle cells, pancreatic beta cells, and neurons. Malfunction of these cells can lead to heart failure, muscular dystrophies, diabetes, pain syndromes, cerebral palsy, paralysis,8 depression and schizophrenia, among many other diseases.” Other recent articles cite applications in pain management, vision challenges such as retinitis pigmentosa and macular degeneration,9,10 and addiction.11 The quality of the light pulse is critical in each of these studies.
The details of the classic optogenetics experiment described in Zhang’s article appear in the caption for Figure 2. This figure encapsulates the essence of optogenetics. First, it demonstrates how a chimeric protein can be created by fusing channelrhodopsin-2 (ChR2), a protein that activates neuronal activity, to a gene for a fluorescent protein (in this case, YFP), used as a reporter for imaging (Figure 2a). Second, it illustrates how a flexible light source can be programmed to photostimulate at various pulse frequencies (Figure 2d). Finally, it reveals the power of optogenetics experiments to image (Figure 2b, scanning confocal microscopy), while simultaneously conducting electrophysiology experiments (Figures 2c, 2d and 2e).
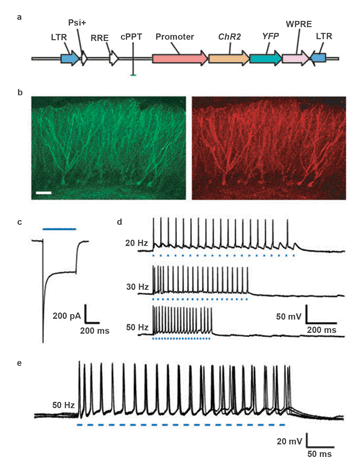
Figure 2. Functional expression of channelrhodopsin 2 (ChR2) in intact rodent hippocampus: (a) Lentiviral vector used for delivery of ChR2 into the brain (LTR = long terminal repeat; RRE = rev response element). ChR2, fused to the gene for a fluorescent protein (XFP; yellow fluorescent protein (YFP) was used here), is driven here by the EF-1 promoter. The HIV-1 central polypurine tract (cPPT) and woodchuck hepatitis post-transcriptional regulatory element (WPRE) also are included for strong long-term expression. (b) Scanning confocal image of dentate gyrus granule cells expressing ChR2-EYFP (yellowish-green fluorescent protein) in the adult mouse hippocampus (left, EYFP fluorescence; right, rhodamine conjugated anti-GFP fluorescence; scale bar, 50 µm). (c) Inward current in a voltage-clamped neuron in an acute slice evoked by 500 ms of 470-nm blue light (indicated by blue bar). (d) Voltage traces showing spikes in a current-clamped hippocampal dentate gyrus hilar interneuron in an acute adult mouse slice evoked by 20-, 30- or 50-Hz trains of light pulses (each blue dash represents one 10-ms light flash). (e) The five traces of 50-Hz spike trains (95 spikes resulting from 100 light flashes) superimposed to demonstrate the low temporal jitter, reliability and sustainability of ChR2-based photostimulation. Traces were collected using a Sutter Lambda DG-4 rapid-wavelength switcher (Chroma HQ470/40× excitation filter, 40× water immersion objective and 300-W xenon lamp, giving rise to ~10 mW/mm2 at the focus) and without the addition of exogenous ATR. Figure reprinted with permission; courtesy of Dr. Feng Zhang et al.7
This experiment was especially important because it was guided by the paper’s co-authors, Dr. Edward S. Boyden and Dr. Karl Deisseroth, both pioneers in optogenetics, and also because the overlay of the five traces in Figure 2e (95 spikes resulting from 100 light flashes) clearly demonstrates the reliability and sustainability of ChR2, now a widely used probe in this field.
Meet the author
Barbara Foster is president and chief strategic consultant of The Microscopy & Imaging Place Inc. and Microscopy/Microscopy Education (MME); e-mail: [email protected].
Acknowledgment
The author wishes to thank the Sutter Instrument staff, especially Dr. Jack H. Belgum, for input on this article, as well as Dr. Richard J. Bookman and Dr. Peter Beemiller for their time and insight.
References
1. Editorial (January 2011). Method of the Year 2010. Nat Meth, Vol. 8, Issue 1. Published online Dec. 10, 2010 (doi: 10.1038/nmeth.f.321).
2. K. Deisseroth (January 2011). Optogenetics. Nat Meth, Vol. 8, Issue 1, pp. 26-29. Published online Dec. 10, 2010.
3. Sutter Instruments, DG-4 Application Note.
4. P. Beemiller et al. Bitplane application note: Tracking T-Cell antigen receptors during formation of immunological synapses. www.bitplane.com/learning/tracking-t-cell-antigen-receptors-during-formation-of-immunological-synapses-immunology.
5. P. Beemiller et al (July 2012). Research paper: Integration of the movement of signaling microclusters with cellular motility in immunological synapses. Nat Immun, Vol. 13, Issue 8, pp. 787-795 (doi: 10.1038/ni.2364).
6. G. Boas (October 2010). Optogenetics: A conversation with Ed Boyden. BioPhotonics, pp. 20-23.
7. F. Zhang et al (October 2005). Channelrhodopsin-2 and optical control of excitable cells. Nat Meth, Vol 3, Issue 10, pp. 785-792.
8. W. Alilain et al (Nov. 12, 2008). Light-induced rescue of breathing after spinal cord injury. J Neuro, Vol. 28, Issue 46, pp. 11862-11870.
9. A. Paddock (March 2012). Optochemical genetics to turn pain off, sight on. BioPhotonics, p. 9.
10. S. Briggs (October 2013). Optogenetics and other neurobiology applications benefit from advances in optics. BioPhotonics, pp. 33-35.
11. (September 2011). Reward-seeking behavior regulated with optogenetics, BioPhotonics, pp. 14-15.