Advancements in instrumentation, along with improvements in data acquisition speed and spatial resolution, have fostered a pump-and-probe technique that resolves infrared chemical images.
BY CÁSSIO LIMA AND ROY GOODACRE, UNIVERSITY OF LIVERPOOL
Due to its unique molecular fingerprinting capability, infrared spectroscopy has been widely used to interrogate the overall biochemistry of biological systems. Over the years, this capability has proved the technique’s worth as a powerful physicochemical method with broad applications within the life sciences. A novel far-field pump-and-probe technique called optical photothermal infrared (O-PTIR) spectroscopy has recently been developed to acquire infrared chemical images from samples quickly. The technique’s impacts and limitations, as well as its future potential in biomedical research, can be projected, along with its potential applications as a tool for single-cell microbial metabolomics and for studying tissue diseases.
Infrared spectroscopy is a technique used to perform chemical analysis based on the absorption of infrared radiation by chemical bonds within a sample. The ability of this vibrational spectroscopic technique to interrogate chemical functional groups gained a lot of attention in the early 1900s when William Weber Coblentz demonstrated in his research that groups of atoms in organic compounds absorb specific wavelengths in the mid-IR range (2500 to 50,000 nm)1. Since Coblentz’s findings were published, advancements in instrumentation and optics have contributed to the development of infrared spectroscopy as an analytical tool in the 20th century.
Courtesy of iStock.com/Mohammed Haneefa Nizamudeen
Beckman released its first commercial infrared spectrometer in 1942, and PerkinElmer unveiled its own infrared spectrometer in 1944. At the time, infrared spectroscopy was already established as a powerful technique for analyzing the chemical composition of organic compounds. However, the infrared spectrometers available were based on dispersive optics such as prisms and gratings, which led to potential thermal degradation of the sample. This configuration had several shortcomings, such as poor repeatability, a slow scanning speed that led to low throughput, and the fact that most of the operations had to be performed manually.
The technical innovation that overcame these operational deficiencies inherent in the traditional design came in the late 1960s and early 1970s, when the third-generation infrared spectrometers that replaced dispersive optics with a Michelson interferometer became commercially available. In an interferometric infrared spectrometer, infrared spectra are generated in the time domain according to the position of the moving mirror placed on one of the arms of the Michelson interferometer. Then a fast Fourier transform is applied to obtain infrared spectra from the interferogram (that is, it converts signal from the time domain to the frequency domain). Fourier transform infrared (FTIR) spectrometers collect infrared spectra in seconds and provide high-quality spectra by signal averaging. To this day, FTIR spectrometers are still among the most popular types of systems used to acquire infrared data from samples.
In 1953, PerkinElmer introduced the first infrared microspectrometer, which combined the chemical specificity integral to infrared spectroscopy with the spatial analysis made possible by a microscope. But despite the novelty of the concept, the system was once again based on dispersive optics, and the concept failed commercially due to technical impracticalities of assembling such a system on a broad scale. Infrared microspectroscopy remained an underused technique until the mid-1980s, when microscopes were combined with FTIR spectrometers, which improved the data acquisition speed and, importantly, the ease of use. The combination of FTIR technology and microscopy enabled the collection of infrared spectra from microscopic regions of a sample, allowing researchers to map the distribution of chemical species in heterogeneous samples.
To expand the range of applications of FTIR microspectroscopy, a variety of optical geometries — such as transmission, reflection, transflection, and attenuated total reflection — have been developed to enable the collection of infrared chemical maps from various types of samples. Currently, FTIR microspectroscopy is a commonly used infrared microscope system in many different laboratories and industries worldwide.
The three major parts of an FTIR microscope are the source, the interferometer, and the detector. Modern FTIR systems are equipped with a source that emits infrared radiation in the mid-IR range, a Michelson interferometer, and a linear and/or two-dimensional array detector. Together, these parts play an important role in establishing spatial resolution in a sample, as well as the scanning speed of chemical maps. The spatial resolution in an FTIR microscope (and any other far-field microscope) is limited by the so-called diffraction limit, which restricts the spatial resolution (the minimum detectable separation between two points that enables them to be distinguished in an image) of a system to the order of the wavelength used for the measurement. Thus, due to the long wavelength of infrared light (λ = 2.5 to 10 μm), a state-of-the-art FTIR microscope provides a spatial resolution of ~10 µm (at 1000 cm−1), therefore limiting the map collection to micrometer-size areas2.
To overcome this constraint, infrared spectroscopy has been coupled with near-field optical techniques — such as atomic force microscopy-infrared (AFM-IR) spectroscopy and infrared scattering-scanning near-field optical microscopy (IR-SNOM) — allowing the collection of spectra at a spatial resolution smaller than the wavelength of infrared light in the nanoscale. However, these additional optical techniques are expensive and laborious, they require good contact between the scanning probe and the sample, and they rely on complex instrumentation and a slow scanning speed compared to an FTIR microscope3.
The rise of O-PTIR
Recently, however, a novel far-field pump-and-probe configuration based on O-PTIR spectroscopy has been developed to quickly acquire infrared chemical maps of samples with submicron spatial resolution. Within this architecture, the pump beam is a tunable pulsed mid-IR quantum cascade laser, while the probe beam is a continuous-wave laser (785 or 532 nm is typical). In O-PTIR spectroscopy, the probe beam measures the photothermal effect induced by the pump beam on infrared-active samples. This system captures the spatial resolution of a sample by using the visible laser as probe beam (~500 nm for 532 nm). This resolution is considerably higher than the resolution provided by an FTIR microscope3. The improvement in spatial resolution brought by the O-PTIR method opens up new opportunities that could not be achieved by earlier iterations of FTIR spectroscopy. The O-PTIR technique is relatively new when compared to traditional FTIR microspectroscopy. The first O-PTIR microscope (called the mIRage and manufactured by Photothermal Spectroscopy Corp.) became commercially available in 2018. Applications of O-PTIR spectroscopy are typically found in the same fields in which FTIR is used, such as in materials science and the life sciences.
O-PTIR in the life sciences
Infrared sensing technology was improved in the late 1990s and early 2000s by the development of focal-plane array detectors, which are essentially highly sensitive thermometers placed in the focal plane. FTIR microspectroscopy gained a lot of attention within the medical community, especially in the field of histopathology, due to its ability to map the distribution of various chemical species in heterogeneous samples. Histopathology and immunohistochemistry remain the gold standard methods for studying the morphological and biochemical changes that occur in cells and tissues during a transition from a healthy state to a diseased state, such as in cancer. These techniques play an important role in diagnosis and treatment as a disease progresses.
However, traditional histopathology techniques are laborious and time-
consuming, and they rely on the experience and knowledge of a pathologist to reach the correct conclusion from the data. These techniques also require expensive labeled antibodies or probes. Thus, the use of FTIR microscopy has potential for tissue imaging because of its ability to provide high contrast between healthy and diseased tissues in an unbiased and label-free manner4. However, FTIR microspectroscopy fails at imaging tightly grouped subcellular structures in a sample due to its poor spatial resolution. This limitation can be overcome by the improved spatial resolution provided by O-PTIR imaging, as illustrated in Figure 15.
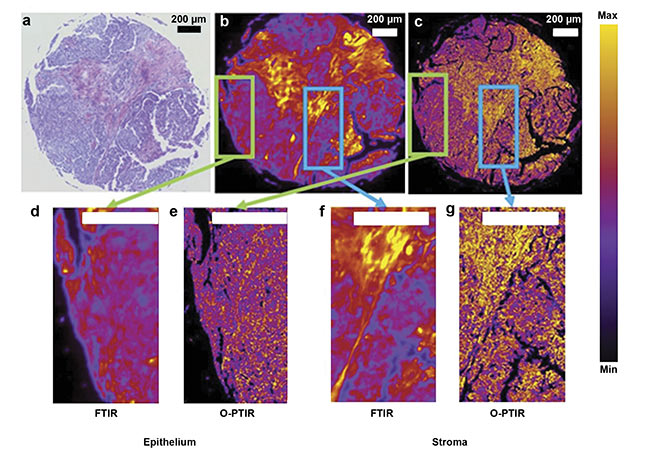
Figure 1. A comparison of images collected at 1650 cm−1 (Amide I, proteins) from the same cancer tissue via Fourier transform infrared (FTIR) and optical
photothermal infrared (O-PTIR) techniques. An optical photomicrograph collected from H&E-stained tissue (a). Chemical maps acquired from cancer tissue core
biopsies via FTIR (b) and O-PTIR (c). Cropped regions around pixels from the same core in FTIR (d, f) and O-PTIR (e, g) images. Adapted with permission from
Reference 5/CC BY 4.0.
The development of the O-PTIR technique has also benefited applications of infrared spectroscopy in microbiology. Many proof-of-concept studies have been published that demonstrate the ability of infrared spectroscopy to study microorganisms such as bacteria and yeast6. However, due to the lack of spatial resolution in an FTIR microscope, most of these studies have focused on assessing bulk microbial populations, and only a few have examined single cells by using AFM-IR and IR-SNOM3, which present several shortcomings and challenges (described above) despite their nanoscale resolution. Considering that most microorganisms are typically about 1 to 2 µm, the spatial resolution provided by O-PTIR imaging is ideal for performing microbial analysis at the single-cell level (Figure 2). Thus, O-PTIR spectroscopy has emerged as a powerful phenotyping tool in single-cell microbial metabolomics, allowing the study of metabolism in whole organisms, as well as cell-to-cell variances.
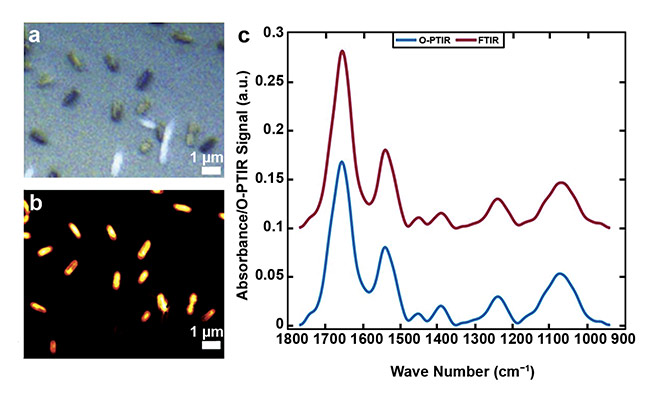
Figure 2. An image acquired from single E. coli bacterial cells (a). A single-frequency image obtained via
O-PTIR at 1655 cm−1 (b). Infrared spectral signatures recorded by FTIR spectroscopy from bulk populations (red line), and from a single bacterial cell recorded by O-PTIR spectroscopy (blue line) (c). Courtesy of
the University of Liverpool.
Another great advantage of O-PTIR over FTIR spectroscopy in life science research is that single-point infrared spectral data acquired through O-PTIR spectroscopy is artifact-free, whereas spectra obtained via FTIR microspectroscopy can exhibit artifacts due to Mie scattering, or the elastic scattered light of particles that have a diameter similar to or larger than the wavelength of the incident light, causing interference2. These dispersive artifacts are commonly seen in spectral data acquired from single biological cells, and they manifest as a sharp decrease in intensity on the high-wave-number side of important absorption bands. This phenomenon results in a shift of the true peak position in collected data, which makes the interpretation of the biochemical features unreliable7.
In addition to achieving submicron spatial resolution and generating artifact-free infrared spectra, some more modern O-PTIR systems can also provide Raman data simultaneously with infrared spectra from the same location at the same resolution. In this arrangement, the visible beam is used as the “infrared” probe, and it is also used as the excitation wavelength to elicit and acquire Raman data2. The use of Raman and infrared spectroscopy together for sample characterization has been well demonstrated, and the two methods are routinely used as complementary analytical techniques to study a wide variety of materials in many situations, such as in forensics and biomedicine. Although both methods provide similar information — such as measuring vibrational modes from molecules — Raman and infrared spectroscopy are performed differently, resulting in advantages and challenges in using them either separately or together.
For example, Raman spectroscopy can be applied to analyze aqueous solutions (Figure 3d), while infrared spectroscopy can only be performed on dry samples. And Raman spectra can be overwhelmed by laser-induced fluorescence (Figure 3b), whereas infrared spectroscopy is fluorescent-free (Figure 3a). Thus, by combining both techniques in a single system, O-PTIR becomes a powerful, versatile tool for characterizing samples, with promising applications in materials science.
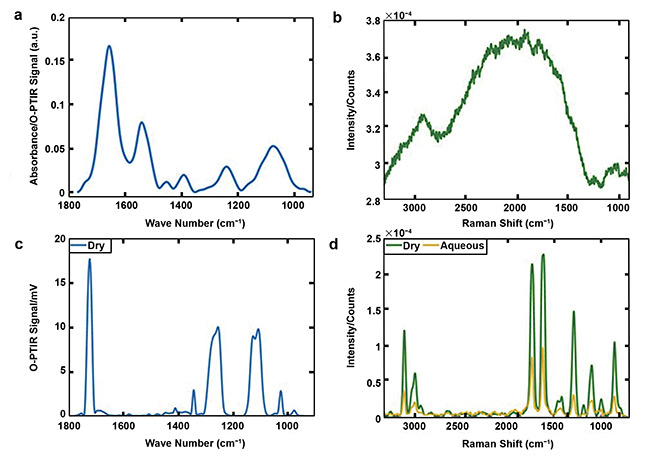
Figure 3. O-PTIR (a) and Raman (b) spectra acquired from the bacterium Bacillus sphaericus. O-PTIR (c) and Raman (green line, d) spectra collected from dry
polyethylene terephthalate (PET), along with the Raman spectrum of PET from an aqueous medium (yellow line, d). Courtesy of University of Liverpool.
More than a century has passed since Coblentz demonstrated the ability of infrared spectroscopy to interrogate chemical functional groups in organic compounds. Since then, advancements in instrumentation — along with the need for improvement in data quality, scanning speed, and spatial resolution — have contributed to the development of technologies that can acquire infrared data from various types of samples.
O-PTIR spectroscopy has emerged as a new infrared technique with several key advantages over classical infrared spectroscopy, such as improved spatial resolution, contactless measurements, simple sample preparation, and the absence of spectral distortions due to scattering. These features will extend the applications of infrared spectroscopy to scientific problems that require analyzing samples at submicron scale. And the technique may in the future provide insights for the development of new far-field infrared technologies with nanoscale spatial resolution.
Meet the authors
Cássio Lima, Ph.D., obtained a Bachelor of Science degree in physics from the Federal University of Uberlândia in Brazil, and a Master of Science degree and doctorate degrees from the University of São Paulo in Brazil. In 2019, he joined professor Roy Goodacre’s group at the University of Liverpool in England, where Lima currently works as a research associate. The main area of his research involves characterization of biological systems via vibrational spectroscopy, including infrared (FTIR and O-PTIR) and Raman spectroscopy (spontaneous Raman, CARS, SRS, and SERS); email: [email protected].
Roy Goodacre is professor of biological chemistry at the University of Liverpool and co-director of the Centre for Metabolomics Research. His research interests fit broadly within analytical biotechnology and systems and synthetic biology. He has helped establish mass spectrometry-based metabolomics for long-term studies and has employed metabolomics for clinical and plant studies, as well as for understanding microbial systems. He has also developed a variety of Raman spectroscopy approaches for bioanalysis, with a particular focus on metabolite quantification and chemical image analysis; email: [email protected].
References
1. A.J. Sommer (2020). Spectroscopy: Introduction to the Techniques and Recent Advances. L. Bush, ed. An e-book from Spectroscopy magazine.
2. M. Kansiz et al. (2020). Optical photothermal infrared microspectroscopy with simultaneous Raman — a new non-contact failure analysis technique for identification of <10 µm organic contamination in the hard drive and other electronics industries. Micros Today, Vol. 28, No. 3, pp. 26-36.
3. C. Lima et al. (2021). Imaging isotopically labeled bacteria at the single-cell level using high-resolution optical infrared photothermal spectroscopy. Anal Chem, Vol. 93,
No. 6, pp. 3082-3088.
4. A. Ogunleke et al. (2017). Fourier-transform vs. quantum-cascade-laser infrared microscopes for histo-pathology: from lab to hospital? TrAC Trends Anal Chem, Vol. 89, pp. 190-196.
5. C. Gajjela et al. (2022). Leveraging mid-infrared spectroscopic imaging and deep learning for tissue subtype classification in ovarian cancer, www.doi.org/10.48550/arxiv.2205.09285.
6. N. AlMasoud et al. (2021). Discrimination of bacteria using whole organism fingerprinting: the utility of modern physicochemical techniques for bacteria typing. Analyst, Vol. 146, pp. 770-788.
7. P. Bassan et al. (2010). RMieS-EMSC correction for infrared spectra of biological cells: extension using full Mie theory and GPU computing. J Biophotonics, Vol. 3,
No. 8-9, pp. 609-620.