Raman systems continue to evolve — from benchtop scientific instrumentsb to portable analytical devices that are used in biomedical applications, with optical filters playing a critical role.
Jason Palidwar, Iridian Spectral Technologies, an IDEX Optical Technologies Company
While Raman spectroscopy has long been useful in pharmaceutical analysis and the identification of hazardous materials, the technology’s usefulness in biomedicine has also been expanding due to less bulky, miniaturized instrumentation. Its inherent capability to distinguish molecular composition is being integrated into specialized diagnostics, in applications such as endoscopy, cancer detection, bone density, and additional in vivo and in vitro biomedical systems.
Raman spectroscopy can be used to noninvasively measure bone density, an indicator of osteoporosis. Courtesy of iStock.com/?UserGI15994093.
Historically, practitioners of Raman spectroscopy have struggled with the elusive nature of Raman scattered “signal” photons from within the Rayleigh scattered source background. Fewer than one in a million photons exchange vibrational energy with the molecules under test and exhibit the associated Raman wavelength shift. To extract this Raman signal from the background, highly wavelength-selective optical filters are necessary to provide more signal, with less background to the detectors to enable the visibility of molecularly unique Raman fingerprint characteristics.
The adaptation of filters has become even more important as the instrumentation has grown smaller. In many cases, these point-of-use portable laboratories and in vivo diagnostic setups have required filters with functionality similar to their benchtop predecessors. The need still exists to clean up the excitation source (laser line filters), to selectively direct the excitation and Stokes scattered light (dichroic mirrors), and to isolate the Raman signal from the excitation background (edge-pass or notch filters). However, in contrast to lab instruments for scientific analysis, portable analytical instruments typically require less precision, because the analysis involves comparing an acquired spectrum to a library database of expected possible spectra. The identification is based on the closest match that is found.
A brief introduction
In Raman spectroscopy, an intense laser beam is used to illuminate a sample under test. Incident light interacts with molecules in the sample, with the challenge of detecting the scarce, inelastically (Raman) scattered photons. The Raman scattered photons are of particular interest because their interaction with vibrational modes of the molecules under test provides “fingerprint” spectra that identify the molecular constituents of the sample (and even the relative concentration of molecules) once processed and measured by a dispersive or Fourier transform spectrometer.
In the past, Raman spectroscopic analysis relied on large, expensive benchtop instruments and existed only in labs in which collected samples were brought in for analysis. Advancements in the miniaturization of sources and detectors, along with improvements to the software and data analysis algorithms, have enabled manufacturers to “take the lab to the field.” Chemical analysis applications in fields such as hazardous materials identification, anti-counterfeiting, analysis of drugs, airport scanning of liquids, and in-process analysis have all flourished consequently. And each of these applications requires filters that meet certain parameters.
Advantages of Raman
There are many advantages to using Raman spectroscopy to identify molecular constituents of samples, be they chemicals, materials, or biological samples, due to the physics involved. Raman is quantitative: The intensity of Raman scattering from a sample is directly proportional to the concentration of molecules present — with high precision, sensitivity, and specificity — and so it provides fingerprint molecular identification and relative content levels.
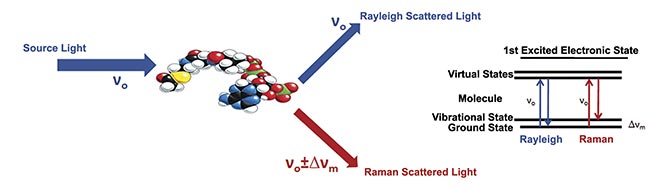
Figure 1. An illustration of the process of Raman scattering. Courtesy of Jason Palidwar/Iridian Spectral Technologies.
• Raman typically does not require sample preparation: Unlike fluorescence analysis, which often requires molecularly specific fluorescence tags or markers to be introduced to the sample to be tested, Raman scattering is a fundamental physical property of the molecules present and so the incident light interacts directly with the unadulterated sample.
• Raman is nondestructive/noninvasive: Raman analysis does not inflict damage or chemical change to the sample being analyzed and can be used in vivo.
• Raman scattering is intrinsic to most biological molecules (i.e., those that undergo a change in polarizability during vibration) but is unaffected by water and can, effectively, “see through” water as the polar bonds in H2O are weak Raman scatterers. This is particularly important for in vivo analysis due to the huge proportion of our bodies that is made up of water (~60%).
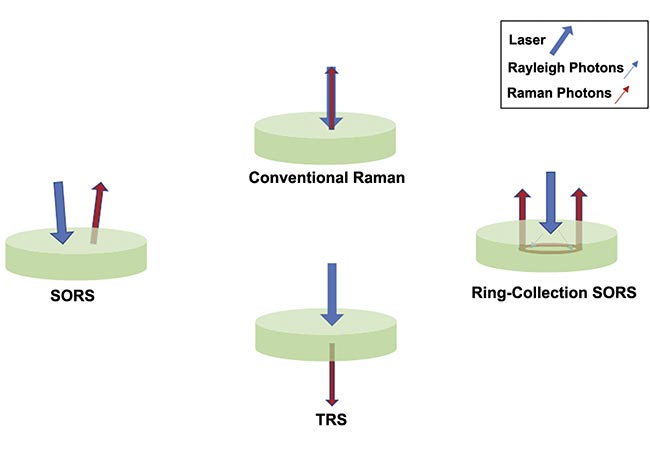
Figure 2. An illustration of the concepts behind spatially offset Raman spectroscopy (SORS) and transmission Raman spectroscopy (TRS). Courtesy of Jason Palidwar/Iridian Spectral Technologies.
Additionally, there are practical instrumentation advantages to Raman analysis. The acquisition of a single Raman spectrum can take <1 s, which provides significant advantages in turbid media or in a living, moving biological sample. And instrumentation can be made to be portable and/or flexible. Raman analytical systems can be configured as stand-alone, hand-held instruments or used with fiber probes connected to benchtop microscopic instruments, and the filter plays an integral role in both.
Conventional backscatter Raman employs illuminating the laser and detector in the same optical path. For opaque, or turbid, media, most Raman photons necessarily come from the sample surface. There is little penetration as deeper Raman photons are absorbed or scattered before getting back to the detector, which, in biological tissues, limits backscatter Raman microscopy to resolving signals within ~100 to 200 µm of the sample surface. There are, however, alternative techniques and approaches to Raman analysis that can overcome some of the challenges of biological materials.
Spatially offset Raman spectroscopy (SORS) relies on a deliberate offset between the incident laser and the collection optics (in one of several different possible configurations). This offsets bias capture of Raman photons from deeper zones in the sample that statistically tend to migrate from the illumination zone (through a “random walk” process), and the depth probed can be stratified by adjusting these spatial offsets.
Transmission Raman spectroscopy (TRS), is a special configuration of SORS in which the illumination and collection optics are on opposite sides of the sample, therefore requiring a use case with access to both sides of the samples. Unlike SORS, TRS cannot be used to select specific stratified layers or penetration depths, but it can acquire a volumetric Raman signal for the entire sample being interrogated.
These two techniques enable the observation of subsurface phenomena to depths from millimeters to tens of millimeters (in the special case of TRS), which is consistent with the needs of in vivo applications seeking to see subcutaneous phenomena or structures.
As mentioned previously, Raman scattering analysis suffers from low signal (less than “one in a million”) and this dearth of signal is exacerbated further in a SORS configuration because there is more scattering in general. Surface-enhanced Raman spectroscopy (SERS), and its spatially offset variant, surface-enhanced-SORS (SESORS), use nanoparticles (often gold in many bio applications) and a reporter molecule to enhance the Raman signal severalfold. SESORS takes advantage of the enhanced Raman signal provided by SERS with the subsurface analysis of SORS. These methods help to mitigate the challenges of low signal but are somewhat invasive in that they require intravenous injection of nanoparticles, but the imaging itself is noninvasive.
Application to bioanalysis
Several medical conditions are accompanied by chemical alterations in associated cells and tissue, and these changes can be detected by the chemical sensitivity and specificity of Raman. Though not yet used in standard medical practices, there are several bioanalytical Raman applications being investigated, and filters will play a key role in bringing the instrumentation into the mainstream.
The holy grail for persons with type 1 diabetes would be noninvasive optical monitoring of blood glucose levels. Work to date has involved SERS/SESORS with implantable detection arrays, which, while not truly noninvasive, may offer improvement compared with current continuous glucose monitoring technologies that require re-insertion every 10 to 14 days1.
Many other ex vivo/in vitro applications are under exploration as well, including the determination of gout and celiac disease, examples in which fluid samples are analyzed ex vivo to diagnose disease. Specialized filters are required for robust and reliable diagnosis.
The role of optical filters
Despite their smaller footprint, these point-of-use portable laboratories are highly dependent on the components they contain, including filters. But portable instruments usually require less precision, since the data they collect is compared to an existing database of available spectra. These instruments can, therefore, be designed for analysis and identification of specific chemical compounds, allowing simplicity in the range and accuracy of sources, detectors, and the filters used for wavelength discrimination.
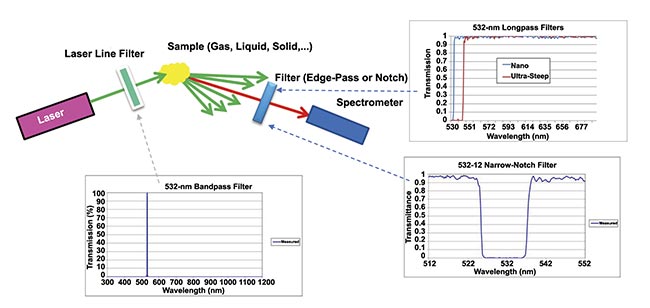
Figure 3. An illustration of data collection with the aid of optical filters in a Raman setup. Courtesy of Jason Palidwar/Iridian Spectral
Technologies.
These differences have allowed optical filter developers to pursue cost efficiency as much as peak performance. Filter manufacturers have the design freedom to sacrifice some spectral finesse for increased manufacturability to yield lower costs and increased usability. For example, wide-angle longpass filters may be a preferred solution for customers’ instruments and devices because they can accommodate wider angle of incidence (AOI) ranges, in some cases up to a total field of view (chief ray AOI plus cone half angle) of 7°. The use of this class of filters can reduce instrumentation, build alignment precision, and allow for less precise control of the angular content in the incident beam, reducing component and assembly complexity and costs. These wide-angle longpass filters can also be designed to provide extended wavelength ranges with high transmission levels (≥95%), with corresponding extended blocking wavelength ranges at the necessary OD6 Rayleigh blocking level over the broad operating ranges of these devices.
The angle-induced wavelength shift associated with any multilayer thin-film optical filter is a fundamental consequence of the physics of interference films. This effect needs to be accounted for and overcome as the data is collected so as not to lose Raman scattered signal and, more importantly, not to introduce Rayleigh scattered noise into the system. However, with some SORS configurations, this angle shift can be put to positive use to enhance the signal from the sample under test, reflecting the scattered Rayleigh light (at non-normal AOI) back into the sample.

Figure 4. An illustration of an off-angle Raman effect. Adapted with permission from Reference 2.
As discussed previously, most incident photons scatter elastically. By positioning a laser bandpass filter in close proximity to the sample under test, many of these elastically backscattered photons will be incident on the backside of the filter at non-normal AOIs2. The angle shift results in many of these backscattered laser photons reflecting off the filter by which they were initially transmitted, sending them back into the sample, and increasing the opportunity for a Raman scattering event. Further enhancement can be obtained in Raman signal by using a shortpass filter or high wave number cut-off longpass filter on the collection side in a TRS configuration to similarly reflect Rayleigh photons back into the sample.
This enhancement could lead to further research and clinical developments in areas such as:
1. Noninvasive determination of bone density in osteoporosis (via SORS). Rather than x-ray or CT (which are expensive and sometimes inaccurate due to the inability to measure collagen), Raman provides low-cost, nonionizing information for both organic and inorganic components of bone (measuring “mineralization” of bone, which is an indicator for fractures)3.
2. In vivo breast cancer screening (via SORS and TRS). The molecular specificity provided by Raman will lead to the reduced need for verifying biopsies compared with mammography and MRI. Additionally, these subsurface Raman analytical techniques may be able to chemically identify differences in benign versus malignant microcalcifications at depths of up to 40 mm, a technique that is advancing into human trials3.
3. Skin cancer lesion detection via SORS3.
4. In vivo brain (glioblastoma) aided by SESORS. This has been demonstrated in mice through intact skulls, employing reporter molecules rather than the fingerprint of the tumors themselves3.
5. Esophageal, gastric, colon, and lung cancer detection via Raman-enabled endoscopy. The determination of tumor margins can be made less subjective by identifying molecular differences between benign and malignant tissue, reducing unnecessary removal of healthy tissue while avoiding incomplete excision of cancerous tissue4.
6. Jermyn et al. at McGill University Brain Tumour Research have developed a hand-held fiber optic Raman probe (operating at 785 nm) with 93.5% sensitivity and 91% specificity in distinguishing between cancerous and benign tissue in real-time brain surgery5.
Meet the author
Jason Palidwar joined Iridian Spectral Technologies in 2006 and is currently product group manager of Aerospace and Specialty Optics, with a focus on optical filters for use in laser-satcom and Earth observation. He has more than 20 years of experience working with thin-film optical filters and has developed filter specifications together with Iridian’s customers to optimize solutions for a wide range of technical and commercial needs in applications such as telecom, Raman spectroscopy, fluorescence microscopy, 3D entertainment, and IR remote sensing. Palidwar has a Master of Science degree in physics from McMaster University; email: [email protected].
References
1. R. Pandey et al. (2017). Noninvasive monitoring of blood glucose with Raman spectroscopy. Acc Chem Res,
Vol. 50, No. 2, pp. 264-272.
2. P. Matousek. (2007). Raman Signal Enhancement in Deep Spectroscopy of Turbid Media. Appl Spectrosc, Vol. 61, No. 8, pp 845-854.
3. F. Nicolson et al. (2021). Spatially offset Raman spectroscopy for biomedical applications. Chem Soc Rev, Vol. 50, No. 1, pp. 556-568.
4. N. Sharma et al. (2016). Raman spectroscopy for the endoscopic diagnosis of esophageal, gastric, and colonic diseases. Clin Endosc, Vol. 49, No. 5, pp. 404-407.
5. M. Jermyn et al. (2015). Intraoperative brain cancer detection with Raman spectroscopy in humans. Sci Transl Med, Vol. 7, No. 274, p. 274ra19.